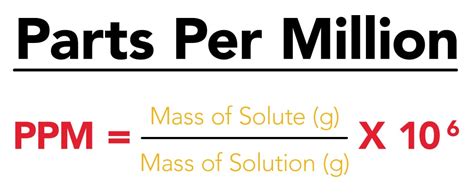Parts Per Million (PPM) is a unit of concentration used to describe very low concentrations of substances, especially useful in scientific and engineering fields. Understanding PPM is crucial for accurate measurements in areas like chemistry, environmental science, and industrial applications. This guide will walk you through everything you need to know about PPM—from its fundamental concepts to practical applications—ensuring you have a solid grasp of how to use and interpret PPM in real-world scenarios.
In fields where precise measurement is key, the concept of Parts Per Million (PPM) provides a valuable way to express minute concentrations. But what exactly does PPM mean? PPM refers to the number of parts of a substance per one million parts of the solution or mixture. This guide aims to demystify PPM, offering practical examples, actionable advice, and solutions to common challenges users face when working with PPM.
Understanding Parts Per Million (PPM): The Basics
PPM is a powerful tool for expressing very small concentrations in a clear and understandable way. It’s especially useful in scientific contexts where substances are present in extremely low quantities, such as:
- Chemical analysis in laboratories
- Environmental monitoring for pollutants
- Quality control in manufacturing
To calculate PPM, you generally use the formula:
PPM = (Part/Total) x 1,000,000
Quick Reference
Quick Reference
- Immediate action item: Always ensure your units are consistent when calculating PPM to avoid errors.
- Essential tip: Convert all measurements to the same units before calculating PPM to maintain accuracy.
- Common mistake to avoid: Overlooking the importance of precise measurement tools which can lead to significant errors in PPM calculations.
Calculating PPM: Step-by-Step Guide
Calculating PPM involves a straightforward yet meticulous process. Here’s how to do it effectively:
- Identify the part and the total: Determine the amount of the substance (the part) and the total amount of the solution or mixture (the total).
- Convert to the same unit: Ensure both the part and the total are measured in the same unit. For instance, if one is in grams and the other in kilograms, convert to kilograms or grams.
- Calculate PPM: Use the formula: PPM = (part/total) x 1,000,000. Plug in your values and compute the result.
- Review for accuracy: Double-check your calculations and ensure that all units are consistent.
For example, if you have 0.002 grams of solute in 2 liters of a solution, convert the volume of the solution to grams (assuming 1 liter of water is 1 gram):
| Step | Calculation |
|---|---|
| Convert liters to grams | 2 liters = 2,000 grams |
| Apply PPM formula | PPM = (0.002 / 2000) x 1,000,000 = 1 PPM |
Real-World Applications of PPM
Understanding PPM is crucial in numerous fields:
- Environmental Science: Measuring pollutants in air or water to assess environmental health.
- Pharmaceuticals: Ensuring the precise concentration of active ingredients in medications.
- Food and Beverage Industry: Monitoring the levels of additives or contaminants.
In environmental science, for instance, PPM can be used to express the concentration of carbon dioxide in the atmosphere. Accurate PPM measurements are essential for understanding climate change impacts and for developing strategies to mitigate pollution.
Detailed How-To: Calculating PPM for Different Scenarios
Here are detailed step-by-step instructions for calculating PPM in different scenarios to ensure you can apply this knowledge in various contexts.
Calculating PPM for Chemical Solutions
When working with chemical solutions, calculating PPM can help you determine the concentration of solutes in a solvent. Follow these steps:
- Identify your components: Determine the mass of your solute and the total volume or mass of your solution.
- Convert if necessary: Ensure that your units are consistent. If you are working with volumes, convert them to mass if needed (e.g., 1 liter of water is approximately 1000 grams).
- Calculate PPM: Use the formula: PPM = (mass of solute/mass of solution) x 1,000,000 for mass/mass solutions or PPM = (volume of solute/total volume of solution) x 1,000,000 for volume/volume solutions.
- Review for accuracy: Double-check your calculations to ensure no mistakes.
Example: You have 0.005 grams of sodium chloride (NaCl) dissolved in 500 grams of water.
| Step | Calculation |
|---|---|
| Apply PPM formula | PPM = (0.005 / 500) x 1,000,000 = 10 PPM |
Calculating PPM for Environmental Samples
Environmental samples often require PPM calculations to determine the concentration of pollutants. Follow these steps:
- Collect sample: Gather your environmental sample, ensuring you have accurate volume or mass.
- Analyze sample: Perform necessary tests to determine the mass or volume of the contaminant in the sample.
- Apply PPM formula: Use the formula: PPM = (mass of contaminant/mass of sample or volume of contaminant/volume of sample) x 1,000,000.
- Review for accuracy: Double-check your calculations and make sure units are consistent.
Example: You have a water sample with 0.0002 grams of lead in 1 liter of water.
| Step | Calculation |
|---|---|
| Apply PPM formula | PPM = (0.0002 / 1000) x 1,000,000 = 0.2 PPM |
Practical FAQ
How can I avoid common mistakes when calculating PPM?
To avoid common mistakes when calculating PPM, always ensure that the units of your parts and total are consistent before plugging them into the formula. Use precise measurement tools to minimize errors. Double-check calculations and verify the correctness of your units. Keeping detailed notes can also help you trace any potential errors back to their source.
What is the significance of PPM in environmental monitoring?
In environmental monitoring, PPM helps quantify the concentration of pollutants in air, water, and soil, which is crucial for assessing environmental health and compliance with regulations. Low PPM values can indicate minimal contamination, while higher values may signal more significant pollution issues that need to be addressed.
Why is PPM important in the pharmaceutical industry?
PPM is vital in the pharmaceutical industry for ensuring the precise concentration of active ingredients in medications, which affects their efficacy and safety. Accurate PPM calculations help manufacturers produce consistent and high-quality pharmaceutical products.
Best Practices for Working with PPM
Here are some best practices to ensure accurate and reliable PPM calculations:
-
<