Navigating the complexities of NH3—whether it functions as a base or an acid—can be quite perplexing, especially if you’re delving into chemistry for the first time. This guide will demystify NH3, providing clear, actionable guidance to help you understand its surprising nature in various contexts.
To start off, ammonia (NH3) is a fundamental molecule in chemistry, and understanding whether it acts as an acid or a base is crucial for grasping its behavior in different environments. At its core, ammonia acts predominantly as a base, but its properties can sometimes lead to confusion. By comprehending its dual nature, you'll be well-equipped to handle any scenario you encounter.
Why Understanding NH3’s Nature Matters
Ammonia’s role as a base is significant in both organic and inorganic chemistry. When NH3 interacts with acids, it donates electrons, thereby neutralizing the acidity. This fundamental behavior is used in industrial processes, such as in the production of fertilizers, where ammonia neutralizes soil acidity. Conversely, when ammonia encounters certain substances, its basic nature can alter reactions, leading to the formation of new compounds.
Clarifying these dynamics ensures precise outcomes in laboratory settings and practical applications. Whether you're a student, a professional chemist, or just someone curious about chemistry, this guide will offer insights that make NH3’s behavior intuitive.
Quick Reference
Quick Reference
- Immediate action item: Conduct pH tests to determine NH3’s state in a specific environment.
- Essential tip: Learn to identify Brønsted-Lowry bases; NH3 accepts protons to form NH4+.
- Common mistake to avoid: Assuming NH3 behaves as a base in all scenarios without verifying its environment.
Understanding NH3’s Basic Nature
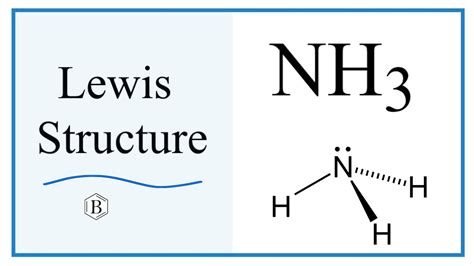
At the heart of ammonia’s behavior lies its molecular structure, consisting of one nitrogen atom and three hydrogen atoms. Its basic nature primarily stems from the nitrogen atom’s lone pair of electrons. This lone pair makes NH3 an excellent candidate for accepting protons (H+), a hallmark of Brønsted-Lowry bases.
Here’s a step-by-step breakdown:
- Identify NH3's molecular structure: nitrogen (N) at the center with three hydrogen (H) atoms attached, forming a trigonal pyramidal shape.
- Focus on nitrogen’s lone pair: nitrogen’s lone pair of electrons is available for donation.
- Proton acceptance: in the presence of an acid, NH3 accepts a proton (H+) to form the ammonium ion (NH4+).
To visualize this reaction, consider the equation:
NH3 + H+ → NH4+
This fundamental reaction underscores NH3’s role as a base in acidic environments.
Detailed Exploration: How NH3 Functions as a Base
To deepen your understanding, let’s explore NH3’s base behavior in detail:
Theoretical Foundations
Ammonia’s basicity is grounded in the Brønsted-Lowry theory, which defines acids and bases by their behavior regarding proton transfer. Here, NH3 exemplifies a Brønsted-Lowry base because it accepts protons easily.
Consider this simplified equation in a basic solution:
NH3 + H2O ⇌ NH4+ + OH-
In this reaction, NH3 accepts a proton from water (H2O) to form NH4+, demonstrating its base capabilities.
Experimental Verification
To confirm NH3’s basic nature experimentally, pH testing is your best tool. When NH3 dissolves in water, it increases the pH level, indicating its basic characteristics.
Follow these steps for an experiment:
- Prepare a solution of NH3 in water.
- Use a pH meter or pH strips to measure the pH level.
- Observe the increase in pH level, confirming NH3’s basicity.
Practical Implications
Understanding NH3’s base behavior has vast implications:
- Industrial applications: In fertilizer production, NH3 neutralizes soil acidity, promoting plant growth.
- Laboratory settings: Knowing NH3’s base nature helps in reactions involving acid-base neutralization.
- Everyday examples: Household cleaners often contain NH3 to clean effectively by neutralizing acidic spills.
Detailed Exploration: When NH3 Can Act in Surprising Ways
While NH3 primarily functions as a base, understanding scenarios where it can exhibit unexpected behavior is vital.
Acidic Environments
In certain specialized conditions, NH3’s behavior can diverge:
When NH3 is subjected to extremely acidic environments or reactions where proton donation is favorable, it may exhibit characteristics that can be puzzling. This doesn’t typically happen under standard conditions but is important to understand in specialized scenarios.
Example scenario: In a highly acidic medium, NH3 can sometimes donate electrons, although this behavior is less common. Recognizing these rare instances aids in understanding the complete spectrum of NH3’s behavior.
Experimental Verification
For verification, you can use specialized equipment to study NH3 in extreme conditions:
- Prepare an ultra-pure acidic solution.
- Introduce NH3 and monitor any deviations from its typical behavior.
- Record any changes in NH3’s activity and compare with standard conditions.
Industrial Context
In industrial settings, conditions can be specially manipulated to exploit NH3’s behavior:
- Specialized synthesis: In certain chemical syntheses, NH3 might be designed to act in ways that deviate from its typical base behavior to achieve specific outcomes.
- Safety measures: Understanding these deviations ensures safety protocols in environments where NH3 is handled under unconventional conditions.
Practical FAQ
What is the primary role of NH3 in acid-base reactions?
The primary role of NH3 in acid-base reactions is that of a base, which means it acts as a proton acceptor. When NH3 comes in contact with an acid, it donates its lone pair of electrons to accept a proton (H+) from the acid, thereby neutralizing the acidity. This base behavior is crucial in various chemical processes, from neutralizing acidic spills in households to large-scale industrial applications.
How can you confirm NH3’s base nature experimentally?
To confirm NH3’s base nature experimentally, you can perform a simple pH test. Dissolve NH3 in water and measure the pH level. In a basic solution, the pH will increase, confirming NH3’s base nature. Additionally, you can observe the reaction where NH3 accepts a proton from water, forming NH4+ and OH- ions, which can be measured with pH indicators or meters.
What happens when NH3 is in an extremely acidic environment?
In extremely acidic environments, NH3’s behavior can sometimes diverge from its typical base characteristics. Though NH3 usually accepts protons, in highly acidic conditions, it might occasionally show deviations by participating in reactions that involve electron donation. This rare occurrence is not typical under standard conditions but is crucial to recognize in specialized industrial applications or experimental setups.
This guide aims to demystify ammonia’s dual nature, ensuring that you are well-equipped to handle it in any context. By following the detailed steps and practical examples provided, you can confidently navigate through its basic behavior and occasionally atypical reactions.
From laboratory experiments to industrial applications, NH3’s behavior is a cornerstone of many chemical processes. Understanding its properties not only helps in academic