Welcome to our comprehensive guide on understanding thermal equilibrium, the fascinating science that governs how heat balances out in various systems. Whether you’re dealing with the warmth of a cup of coffee cooling on your kitchen table or the sophisticated temperature controls in a scientific lab, thermal equilibrium is the principle that underlies these everyday phenomena. This guide aims to give you a solid grasp of the concept through practical, real-world examples, actionable advice, and a problem-solving focus that addresses common user pain points.
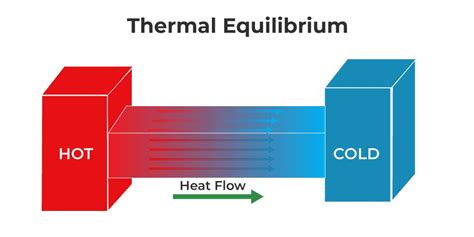
Thermal equilibrium occurs when two or more objects in direct contact reach the same temperature. This equilibrium signifies that no net heat energy is flowing between the objects; they are in a state of balance. It’s crucial to understand this concept, especially when dealing with temperature regulation in both everyday life and specialized scientific contexts. For instance, understanding how to maintain the perfect baking temperature in your oven or how to properly store medications that require specific temperature conditions is essential. This guide will walk you through the key principles of thermal equilibrium, providing practical tips and solutions to ensure you can apply this knowledge effectively.
Step-by-Step Guidance to Achieving Thermal Equilibrium
Here's a straightforward approach to help you navigate the complexities of thermal equilibrium. We will cover immediate action items, essential tips, and common pitfalls to avoid.
Quick Reference
- Immediate action item with clear benefit: Measure the temperature of your objects before and after contact to verify if they have reached thermal equilibrium.
- Essential tip with step-by-step guidance: To achieve thermal equilibrium, ensure there’s sufficient contact time between the objects, and avoid insulating materials that could disrupt heat transfer.
- Common mistake to avoid with solution: Don’t assume equilibrium after a short time. Always allow enough time for heat to distribute evenly, which could take minutes to hours depending on the objects’ size and thermal mass.
Understanding Heat Transfer Mechanisms
To deeply understand thermal equilibrium, it’s vital to know how heat moves between objects. Heat transfer can happen through three primary mechanisms: conduction, convection, and radiation.
Conduction
Conduction is the process where heat energy is directly transferred through contact between materials. Metals are excellent conductors because of their free electrons. For example, when a metal spoon is placed in a hot pot of soup, heat quickly flows from the hot soup to the cooler spoon.
Here’s how to ensure effective conduction:
- Maintain continuous contact between the objects.
- Choose materials with high thermal conductivity.
- Allow sufficient time for the heat to transfer.
Convection
Convection occurs in fluids (liquids and gases) where warmer, less dense regions move and mix with cooler, denser regions. This movement distributes heat throughout the fluid. An everyday example is boiling water: the heat at the bottom of the pot rises as it warms up, creating convection currents.
For effective convection:
- Ensure fluid is in motion to help distribute heat evenly.
- Avoid covering containers completely to let hot areas rise and cooler areas sink.
- Use stirring mechanisms for controlled convection in cooking or heating liquids.
Radiation
Radiation is the transfer of heat through electromagnetic waves without needing a medium. This is how the sun warms the Earth. Materials like metals or water absorb and re-radiate this energy.
Tips for managing radiation:
- Use reflective surfaces to redirect heat where needed.
- Avoid blocking radiation sources when warming or cooling objects.
- Consider thermal insulators to minimize unwanted radiation transfer.
Achieving Equilibrium in Cooking
Understanding thermal equilibrium can vastly improve your cooking and baking results. Ensuring that your oven and ingredients reach the right temperature before starting your recipe is crucial. Here’s a detailed guide on achieving thermal equilibrium in cooking:
Before you start, always preheat your oven to the required temperature to ensure that it reaches thermal equilibrium with the baking tray or dish you’ll use. Here's a step-by-step method:
- Preheat your oven to the required temperature (e.g., 350°F or 175°C).
- Place an oven thermometer inside to verify the actual temperature.
- Wait until the oven stabilizes at the desired temperature before placing your food inside.
- Ensure the baking dish or tray is also at room temperature initially, as sudden temperature changes can affect the cooking process.
- After placing your food in, periodically check with an oven thermometer to ensure it maintains the equilibrium throughout the cooking time.
To prevent common mistakes:
- Don’t open the oven door frequently as it lets heat escape, disrupting equilibrium.
- Avoid using aluminum foil to cover food until you’ve reached the halfway point of the cooking time, as it can trap heat and potentially cause overcooking.
- Make sure your ingredients are at room temperature before baking to help even cooking.
Maintaining Equilibrium in Scientific Labs
In scientific laboratories, maintaining thermal equilibrium is often critical for accurate experiments. Whether you’re calibrating an instrument or preparing a sample for analysis, understanding thermal dynamics is crucial.
- Before conducting experiments, ensure that all lab equipment and samples reach thermal equilibrium with the ambient environment.
- Use a thermal equilibrium chamber if you need to control and stabilize temperature precisely.
- Regularly calibrate your thermometers and ensure they’re placed in the correct position to get accurate readings.
To avoid common lab mistakes:
- Avoid insulating lab equipment as it could lead to inaccurate readings.
- Don’t expose equipment to temperature fluctuations that could affect readings.
- Regularly maintain and calibrate instruments to ensure they are functioning correctly.
Practical FAQ
How can I ensure my room maintains a comfortable temperature all day?
Maintaining a consistent room temperature involves balancing heating and cooling systems efficiently. Here’s what you can do:
- Set thermostats to a moderate temperature and adjust based on occupancy.
- Seal leaks and insulate windows to prevent heat loss during winter and heat gain during summer.
- Use curtains to block direct sunlight in summer and keep the room warm in winter.
- Regularly check heating and cooling systems to ensure they are working efficiently.
- Use fans wisely: circulating air can help in cooling rooms without compromising thermal equilibrium.
Understanding and applying the principles of thermal equilibrium can significantly enhance your cooking, scientific experiments, and everyday temperature management. By following the steps and tips provided in this guide, you’ll be better equipped to achieve and maintain balance in heat across various contexts. Whether it’s in the kitchen or the lab, mastering thermal equilibrium is a valuable skill that brings practical benefits to your daily life.