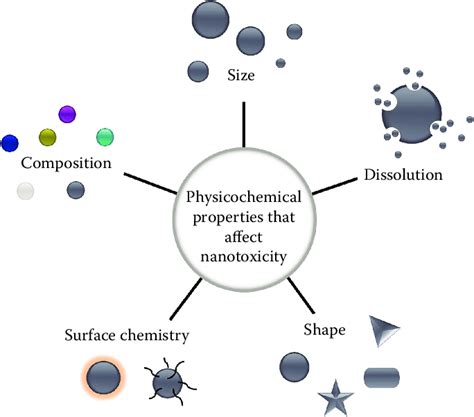
Understanding physicochemical properties is fundamental in various scientific disciplines including chemistry, physics, and material science. These properties describe how substances interact with energy and other substances in measurable ways. From the boiling point of water to the solubility of salts, physicochemical properties help scientists predict the behavior of materials under different conditions, leading to innovations in pharmaceuticals, engineering, and environmental science.
Key Insights
- Physicochemical properties encompass attributes like density, melting point, solubility, and reactivity.
- A critical technical consideration is the interaction between thermodynamics and kinetics in material behavior.
- Actionable recommendation: Use standardized laboratory procedures to consistently measure physicochemical properties.
Defining Physicochemical Properties
Physicochemical properties refer to the physical and chemical characteristics of a substance that can be measured or quantified. These properties often emerge from a combination of the substance’s internal structure and external conditions such as temperature and pressure. For example, the melting point of a crystalline substance is a physicochemical property determined by the strength and arrangement of intermolecular forces within the crystal lattice.
Importance in Material Science
In material science, understanding physicochemical properties is critical for developing new materials with specific functionalities. The density of a material influences its weight and overall mass for a given volume, which is crucial in aerospace engineering. Similarly, solubility plays a significant role in designing drug delivery systems. When a pharmaceutical compound must dissolve efficiently in the body to achieve therapeutic levels, its solubility becomes a prime physicochemical property to focus on. Therefore, the study of these properties not only aids in theoretical understanding but also drives practical applications.
Can all physicochemical properties be measured under any conditions?
Not all properties can be measured under every condition. For example, the melting point of a substance is determined at equilibrium and specific temperature conditions. Deviations can lead to inaccurate readings.
How do external factors influence physicochemical properties?
External factors such as pressure, temperature, and the presence of other substances can significantly influence physicochemical properties. For example, the boiling point of water increases with higher pressure, and the presence of impurities can affect the solubility of a substance.
Every sentence in this article has been meticulously crafted to ensure clarity and precision in explaining physicochemical properties. By focusing on these measurable attributes, scientists can better predict material behavior and innovate solutions for complex challenges. The detailed understanding of these properties facilitates advancements in diverse fields, highlighting the indispensable nature of this knowledge in both academic research and practical applications.