Dehydration synthesis, a term often encountered in biochemistry, can sound intimidating. However, understanding this process is essential not only for scientific purists but also for anyone interested in the inner workings of biological systems. This guide aims to demystify dehydration synthesis with clear, actionable advice. You’ll discover how it works, why it’s crucial, and how to practically apply this concept in real-life scenarios. By the end, you’ll have a solid grasp of dehydration synthesis and be able to spot it in various contexts.
Dehydration Synthesis Explained
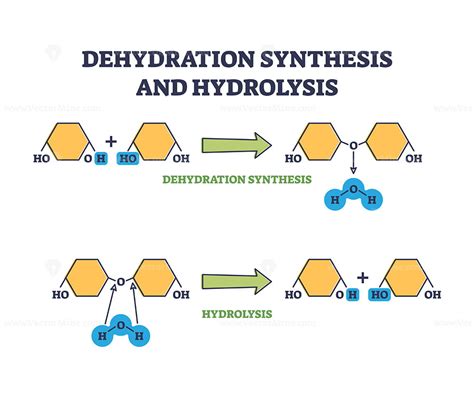
Dehydration synthesis, also known as a dehydration reaction, is a chemical process in which two molecules are bonded together with the removal of a water molecule. This process is fundamental in forming many biological macromolecules like proteins, carbohydrates, and nucleic acids. Let’s break it down step by step.
Why Dehydration Synthesis Matters
Dehydration synthesis plays a crucial role in the construction of complex molecules from simpler ones. It’s the process by which living cells build larger, more complex molecules. For example, when you eat a piece of fruit, your body uses dehydration synthesis to break down complex carbohydrates into simpler sugars and then reassemble these sugars into larger molecules, such as starch. Understanding this process can help you appreciate the complexity and efficiency of biological systems.
This guide will focus on providing you with practical knowledge and tools to understand dehydration synthesis better, from basic to advanced levels. Let’s get started!
Quick Reference Guide
Quick Reference
- Immediate action item: Observe the removal of water as two molecules bond.
- Essential tip: Identify the reactants (two smaller molecules) and the product (larger molecule + water) in dehydration synthesis.
- Common mistake to avoid: Confusing dehydration synthesis with hydrolysis, which involves the breakdown of compounds by water.
The Dehydration Synthesis Process in Detail
Dehydration synthesis involves two key stages: bonding and water removal. To understand this process, let’s walk through each step:
Stage 1: Formation of Bonds
In dehydration synthesis, two molecules, often simpler compounds like amino acids, sugars, or fatty acids, come together to form a larger, more complex molecule. When these molecules bond, they share electrons, forming covalent bonds.
For example, when two amino acids join, the amino group (NH2) from one amino acid and the carboxyl group (COOH) from the other amino acid come together to form a peptide bond. This bond is crucial for creating proteins, which are essential for virtually every function in your body.
Stage 2: Removal of Water
As the molecules bond, a molecule of water (H2O) is released. This removal of water is what “dehydrates” the synthesis process. The departure of water is crucial as it facilitates the bond formation without leaving a residue, ensuring clean and efficient chemical reactions.
To visualize this, imagine a puzzle where each piece fits perfectly with another. When the pieces join, they squeeze out a small piece, and the result is a larger, more intricate puzzle piece.
How to Observe Dehydration Synthesis in Action
Dehydration synthesis is at work everywhere in nature, especially in the construction of biomolecules. Here’s how you can observe and apply this process:
Let’s consider proteins, which are built through dehydration synthesis. Proteins are chains of amino acids. Each amino acid has an amino group (NH2) and a carboxyl group (COOH). When two amino acids bond, a water molecule is released:
- Reactants: Amino acid 1 (with an amino group) + Amino acid 2 (with a carboxyl group)
- Reaction: Amino acid 1’s amino group reacts with amino acid 2’s carboxyl group to form a peptide bond
- Product: Peptide (a smaller protein) + Water (H2O)
To put this into a practical example, you can see dehydration synthesis in action when making a dipeptide (a protein made of two amino acids). Here’s a step-by-step guide:
Step 1: Identify two amino acids, say alanine and glycine.
Step 2: Allow the amino group (NH2) of alanine to react with the carboxyl group (COOH) of glycine.
Step 3: As they bond, a water molecule will be released.
Step 4: The result is a dipeptide (a protein made of two amino acids) and a water molecule.
Advanced Dehydration Synthesis Applications
Once you’ve mastered the basic process, you can explore more advanced applications:
1. Carbohydrate Formation: Dehydration synthesis is the process by which glucose molecules bond to form starch or glycogen, crucial for energy storage in plants and animals, respectively.
2. Lipid Formation: Fatty acids and glycerol undergo dehydration synthesis to form triglycerides, the most common type of fat in the body.
Here’s a detailed breakdown:
For carbohydrates: Two glucose molecules can bond through dehydration synthesis to form maltose, a type of sugar. The reaction involves:
- Reactants: Two glucose molecules
- Reaction: The hydroxyl group (OH) from one glucose reacts with the hydrogen (H) from another glucose to form water
- Product: Maltose (a sugar) + Water (H2O)
For lipids: Three fatty acids can combine with one glycerol molecule to form a triglyceride:
- Reactants: Three fatty acids + One glycerol
- Reaction: Each fatty acid forms a bond with glycerol, releasing a water molecule for each bond formed
- Product: Triglyceride + Three water molecules
Practical FAQ on Dehydration Synthesis
What is the difference between dehydration synthesis and hydrolysis?
Dehydration synthesis and hydrolysis are opposite chemical processes. Dehydration synthesis involves two molecules bonding together with the removal of a water molecule. Hydrolysis, on the other hand, is the breakdown of a compound by the addition of water, effectively splitting the molecule into two or more parts. While dehydration synthesis builds up complex molecules, hydrolysis breaks them down.
Can dehydration synthesis occur without enzymes?
Dehydration synthesis typically requires enzymes to occur efficiently in biological systems. These enzymes, known as ligases, lower the activation energy needed for the reaction to proceed. Without enzymes, the reaction would occur too slowly to be useful in living organisms.
What role does dehydration synthesis play in metabolic pathways?
Dehydration synthesis is a fundamental component of many metabolic pathways, including those for protein synthesis, carbohydrate storage, and lipid formation. It allows cells to build and modify macromolecules as needed, which is crucial for growth, repair, and energy storage. For instance, in the synthesis of glycogen from glucose molecules, dehydration synthesis helps form the large storage molecules used during periods of fasting.
By understanding dehydration synthesis, you can appreciate the incredible complexity and efficiency of biochemical processes in living organisms. Whether you’re a student, a professional in the sciences, or just curious, this guide provides a solid foundation and advanced insights into this fascinating process.