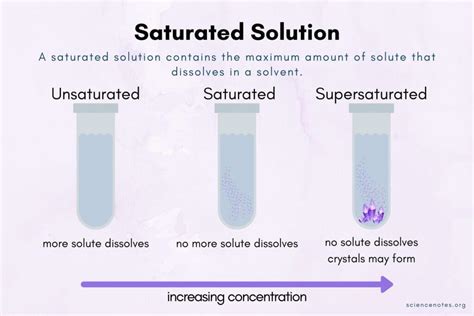
Understanding a saturated solution is essential for anyone involved in fields ranging from chemistry to food science to everyday home experiments. When we talk about saturation in chemistry, we refer to a solution where the solvent has dissolved as much solute as it can at a given temperature and pressure. Beyond this point, any additional solute will not dissolve but will instead remain as a solid at the bottom of the container.
The quest to grasp the concept of saturation often leads to frustration when it seems too abstract or complex. This guide will help demystify this concept by offering practical, actionable advice and real-world examples to cement your understanding. By the end of this guide, you will be able to identify, create, and even troubleshoot issues related to saturated solutions confidently.
Understanding the Basics of Saturated Solutions: A Primer
A saturated solution is one where no more solute can dissolve in the solvent at a particular temperature. This state occurs when the rate of dissolution equals the rate of precipitation. Here’s why this concept is crucial and some common scenarios where it plays a role:
- In pharmaceuticals, knowing when a solution is saturated can help determine the effectiveness of the medication.
- In cooking, understanding saturation can influence the concentration of flavors extracted from spices and herbs.
- In environmental science, saturation levels help predict the behavior of pollutants in water bodies.
To gain a practical grasp on what constitutes a saturated solution, here’s a quick reference:
Quick Reference
- Immediate action item: If a sugar solution doesn’t dissolve any more sugar, it’s likely saturated.
- Essential tip: Stirring the solution at different temperatures can help in dissolving more solute or identifying saturation levels.
- Common mistake to avoid: Assuming that a clear solution means it is unsaturated; clarity does not guarantee saturation.
How to Identify a Saturated Solution: A Step-by-Step Approach
To determine if a solution is saturated, we must employ a series of observations and tests. Here’s a step-by-step process:- Observe the solution: A clear solution may suggest saturation, but it’s not definitive. Look for any undissolved particles or crystals at the bottom.
- Perform a temperature test: Heat the solution and then cool it. If the solute crystallizes upon cooling, it indicates saturation.
- Conduct a saturation test: Add a small amount of extra solute. If it doesn’t dissolve, the solution is saturated.
For instance, when preparing tea, if you add sugar and it doesn’t dissolve, that tea solution is saturated with sugar at that temperature.
How to Prepare a Saturated Solution: Detailed Instructions
Creating a saturated solution involves precise control over the amounts of solute and solvent, temperature, and time. Here’s a detailed guide:
- Choose your solute and solvent: For example, sugar and water, or potassium nitrate and water.
- Dissolve the solute: Gradually add the solute to the solvent while stirring constantly. Continue adding until no more solute can dissolve.
- Assess the saturation: Use the tests mentioned earlier to confirm saturation.
- Control temperature: The saturation point varies with temperature. Keep the solution at a constant temperature using a water bath.
- Refrigerate if necessary: Some solutes may precipitate out at lower temperatures, confirming the saturation point.
Consider an example with potassium nitrate:
In a beaker, mix 100 grams of potassium nitrate with 100 mL of water. Stir until no more crystals dissolve. Place the beaker in a water bath set at 30°C. Once complete, remove from heat and let it cool. If the solution doesn’t dissolve additional crystals upon cooling, you’ve created a saturated solution.
Troubleshooting Saturated Solutions: Common Pitfalls and Solutions
When working with saturated solutions, several common issues can arise:
- Incomplete dissolution: Ensure proper stirring and sufficient time for dissolution. Sometimes heating and slow cooling can help.
- Over-saturation: Too much solute may lead to immediate precipitation. Dissolve smaller amounts of solute gradually.
- Temperature fluctuations: Always maintain a constant temperature to avoid spontaneous crystallization.
For example, if you accidentally oversaturated your solution, simply reheat it to dissolve excess solute and start again.
What happens if I keep adding solute to a saturated solution?
When a solution is saturated, adding more solute will result in undissolved solute sitting at the bottom of the container. The solution remains at the same saturation level.
How can I ensure my solution is accurately saturated?
To ensure accuracy, use temperature control, conduct a dissolution test by adding extra solute, and monitor for any precipitation. Stirring at different temperatures can also be beneficial.
Can a saturated solution become unsaturated again?
Yes, by changing the temperature or adding more solvent, a saturated solution can become unsaturated. For instance, heating can increase the solvent’s capacity to dissolve more solute.
This guide should equip you with the knowledge to tackle saturated solutions across various fields confidently. From simple kitchen experiments to advanced scientific research, understanding saturation can enhance precision and effectiveness in diverse applications.