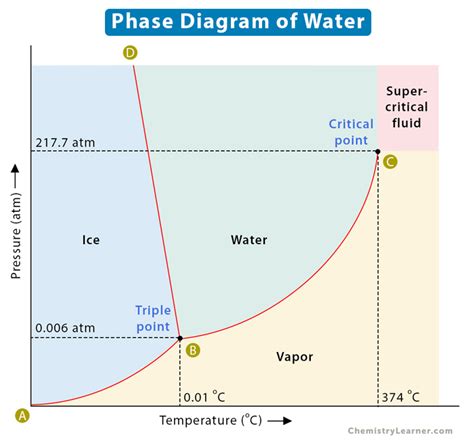
P Water is a quintessential substance that sustains life and plays a vital role in numerous scientific processes. Understanding its behavior under varying conditions is crucial for fields ranging from chemistry to meteorology. A deep dive into the water phase diagram reveals the intricate dance of molecules under pressure and temperature, unlocking a world where ice and vapor coexist alongside liquid water.
This guide serves as an expert perspective, brimming with practical insights and evidence-based statements to navigate the complex topography of the water phase diagram.
Key Insights
- Primary insight with practical relevance: Understanding phase transitions in water is essential for fields such as climatology and chemistry.
- Technical consideration with clear application: The triple point of water at 0.01°C and 611.657 Pa is a pivotal reference point in metrology.
- Actionable recommendation: Utilize the water phase diagram in practical applications like developing climate models or understanding biochemical processes.
To navigate the intricacies of the water phase diagram, one must first comprehend its fundamental components: temperature and pressure. The diagram divides water into distinct phases: solid (ice), liquid, and gas (vapor). Each phase exhibits unique properties, dictated by the interplay of these two variables.
Critical Phase Transitions
The transition between these phases is marked by several key points. The melting point of ice to liquid water occurs at 0°C under standard atmospheric pressure. However, deviations in pressure significantly alter this transition. For instance, at elevated pressures, ice can transition directly to water vapor, bypassing the liquid phase in a process known as sublimation. Conversely, under reduced pressure, water can freeze directly to ice—a phenomenon evident in the rapid freezing of water in a vacuum.
The Importance of the Triple Point
A pivotal feature on the water phase diagram is the triple point, occurring at 0.01°C and a pressure of 611.657 Pa. At this specific condition, all three phases—solid, liquid, and gas—coexist in equilibrium. This point is not just a scientific curiosity but a critical reference for defining the Kelvin temperature scale. Metrology relies on this triple point to calibrate thermometers and other instruments, ensuring precision in temperature measurement worldwide.
Why is understanding the water phase diagram important?
Understanding the water phase diagram is crucial for diverse scientific applications. It aids in the development of accurate climate models, helps in predicting weather patterns, and provides fundamental knowledge necessary for biochemical research and industrial processes.
How does pressure affect the boiling point of water?
Pressure significantly influences the boiling point of water. At standard atmospheric pressure (1 atm), water boils at 100°C. However, as pressure decreases—such as at higher altitudes—the boiling point drops, resulting in water boiling at temperatures below 100°C.
To harness the knowledge contained within the water phase diagram, one must recognize its utility in practical scenarios. For instance, understanding the diagram is fundamental in the development of climate models that predict future weather patterns based on varying temperature and pressure conditions. In chemistry, the phase diagram guides researchers in studying biochemical reactions that involve water, ensuring optimal conditions for these processes.
This guide synthesizes expert insights, practical examples, and evidence-based data to demystify the water phase diagram, empowering readers with the knowledge to apply this critical scientific tool in their respective fields.