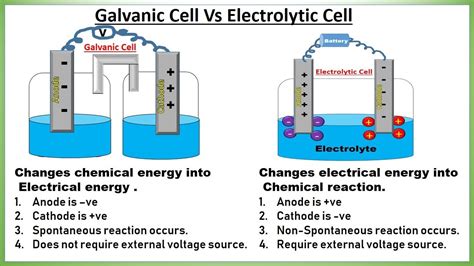
Voltaic cells, also known as galvanic cells, generate electricity by harnessing spontaneous redox reactions. Conversely, electrolytic cells require an external electrical source to force a chemical reaction to occur. Understanding the differences between these two types of cells is essential for applications ranging from batteries to industrial processes. This guide will provide step-by-step guidance to help you comprehend these concepts deeply and apply them practically in real-world scenarios.
The Problem: Choosing the Right Cell for Your Application
One of the common challenges users face is deciding whether to use a voltaic or an electrolytic cell for a particular application. Each type of cell serves different purposes and has distinct advantages and limitations. Misjudging the cell type can lead to inefficiency or even failure in a project. This guide aims to clear up any confusion and arm you with the knowledge to make an informed choice, ensuring that you harness the power that best suits your needs.
Quick Reference
Quick Reference
- Immediate action item: Identify the nature of your application. Is it a naturally occurring redox reaction, or does it require an external power source?
- Essential tip: For applications requiring continuous power supply, a voltaic cell (like a battery) is often more suitable. For processes needing a non-spontaneous reaction, an electrolytic cell is essential.
- Common mistake to avoid: Using a voltaic cell in a scenario that demands external power input or vice versa. Always align the cell type with the reaction’s spontaneity and the energy source availability.
Understanding Voltaic Cells
Voltaic cells, named after Alessandro Volta, generate electrical energy through spontaneous redox reactions. These cells convert chemical energy directly into electrical energy, which makes them fundamental in creating batteries.
Here’s a step-by-step guide on how voltaic cells work and how to implement them effectively:
Step 1: Identify the Components
A typical voltaic cell consists of:
- Two electrodes (anode and cathode)
- An electrolyte that facilitates ion flow
The spontaneous reaction occurs when the anode undergoes oxidation, and the cathode undergoes reduction, generating electrical energy.
Step 2: Setting Up a Voltaic Cell
To set up a voltaic cell, follow these steps:
2.1: Choose appropriate materials. For example, zinc and copper can form a basic voltaic cell.
2.2: Place the anode (zinc) in an electrolyte solution (e.g., sulfuric acid).
2.3: Insert the cathode (copper) in the same solution, ensuring it doesn’t touch the anode.
2.4: Connect the electrodes with a wire to complete the circuit.
Step 3: Measuring Voltage
Once set up, measure the voltage produced using a voltmeter. The difference in electrical potential between the anode and cathode is the voltage of the cell.
Step 4: Using Voltaic Cells
Voltaic cells are widely used in:
- Batteries for portable electronic devices
- Fuel cells for renewable energy
- Galvanic corrosion protection
To ensure longevity, avoid over-discharging the cell and store it at a stable temperature.
Understanding Electrolytic Cells
Electrolytic cells require an external electrical power source to drive a chemical reaction that does not occur spontaneously. These cells are pivotal in electroplating, electrolysis of water, and many industrial processes.
Here’s how electrolytic cells work and how to effectively implement them:
Step 1: Identify the Components
An electrolytic cell includes:
- Two electrodes (anode and cathode)
- An electrolyte that allows ionic movement
The non-spontaneous reaction occurs when an external power source provides the necessary energy to drive the reaction.
Step 2: Setting Up an Electrolytic Cell
Follow these steps to set up an electrolytic cell:
2.1: Select appropriate materials. For example, silver and chloride in a salt solution can form an electrolytic cell.
2.2: Place the anode (silver) and cathode (typically graphite) in an electrolyte solution.
2.3: Connect the electrodes to an external power source such as a battery or power supply.
2.4: Ensure proper polarity to avoid any undesired reactions.
Step 3: Observing the Reaction
With the external power source connected, the anode and cathode will undergo oxidation and reduction respectively, resulting in the desired chemical reaction.
Step 4: Utilizing Electrolytic Cells
Electrolytic cells are crucial in:
- Electroplating to coat metals
- Water electrolysis for hydrogen production
- Refining metals and purifying substances
To maximize efficiency, use high-purity electrolytes and ensure constant monitoring of the cell's operation.
Practical FAQ
What are the differences between a voltaic cell and an electrolytic cell in terms of energy input?
A voltaic cell generates electricity from spontaneous chemical reactions, converting chemical energy into electrical energy without needing an external power source. An electrolytic cell, on the other hand, uses an external power source to drive non-spontaneous chemical reactions. This means a voltaic cell produces power through natural processes, while an electrolytic cell requires an input to force reactions to occur.
How can I determine if I need a voltaic or electrolytic cell for my project?
To determine whether a voltaic or electrolytic cell is needed, consider the nature of the chemical reactions required for your project. If your project relies on natural, spontaneous redox reactions to generate electricity, a voltaic cell is appropriate. If it requires forcing chemical reactions with external energy, an electrolytic cell is the right choice. Additionally, consider the energy source availability and the nature of the output required for the task.
Can I convert a voltaic cell into an electrolytic cell?
No, a voltaic cell cannot be directly converted into an electrolytic cell because their mechanisms are fundamentally different. A voltaic cell relies on spontaneous reactions to produce energy, while an electrolytic cell requires an external power source to drive reactions. However, you can repurpose components from a voltaic cell for use in an electrolytic setup by appropriately modifying the circuit and adding necessary external components.
By understanding these differences and how to implement both types of cells, you can choose the right technology for your specific needs, ensuring efficient and effective outcomes in your projects.