Finding the molar mass of sodium phosphate might seem daunting at first, but with the right approach, it becomes a straightforward process. The molar mass of a compound is essential for various scientific applications, from stoichiometry in chemistry to calculating doses in pharmacology. Let’s delve into the problem-solution approach, providing practical guidance and actionable tips to ensure you understand and can accurately calculate the molar mass of sodium phosphate.
When dealing with the molar mass of a compound like sodium phosphate (Na₃PO₄), it's essential to approach it step-by-step. This task not only aids in understanding chemical compositions but also forms the foundation for more complex chemical calculations. Whether you're a student working on a lab report or a professional in the field needing precise calculations for formulations, knowing how to find the molar mass efficiently can save time and reduce errors. Here, we'll guide you through a process that breaks down this task into manageable steps, providing real-world examples to illustrate each part of the calculation.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Start by writing down the chemical formula for sodium phosphate, Na₃PO₄. This ensures you have all the elements you need to calculate the molar mass.
- Essential tip with step-by-step guidance: Break down the calculation into smaller parts: find the atomic masses of each element, then sum them up according to the formula.
- Common mistake to avoid with solution: Ensure you’re using the most current atomic masses. A common error is using outdated atomic masses, leading to incorrect molar mass calculations.
Understanding the Basics: What is Molar Mass?
Molar mass, often expressed in grams per mole (g/mol), is a measure of the mass of one mole of a given substance. It’s a crucial concept in chemistry, allowing scientists to quantify and compare the amounts of different substances. To calculate the molar mass of a compound, you need to add up the atomic masses of all the atoms in the molecular formula of the compound.
Step-by-Step Guide to Calculate Sodium Phosphate Molar Mass
Calculating the molar mass of sodium phosphate involves a few clear, logical steps. Here’s how you can approach it:
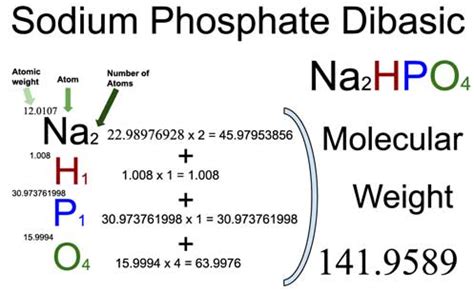
- Identify the chemical formula: The chemical formula for sodium phosphate is Na₃PO₄. This means one molecule of sodium phosphate contains three sodium (Na) atoms, one phosphorus (P) atom, and four oxygen (O) atoms.
- Find the atomic masses: Look up the atomic masses of each element in the periodic table. As of the latest update, the atomic masses are approximately:
- Sodium (Na): 22.99 g/mol
- Phosphorus (P): 30.97 g/mol
- Oxygen (O): 16.00 g/mol
- Calculate the molar mass: To find the molar mass of Na₃PO₄, you'll sum the masses of all the atoms according to their quantities in the formula.
- Multiply the atomic mass of sodium by three (for the three sodium atoms): 22.99 g/mol * 3 = 68.97 g/mol
- Multiply the atomic mass of phosphorus by one (for the one phosphorus atom): 30.97 g/mol * 1 = 30.97 g/mol
- Multiply the atomic mass of oxygen by four (for the four oxygen atoms): 16.00 g/mol * 4 = 64.00 g/mol
- Add these together to get the total molar mass of sodium phosphate: 68.97 g/mol + 30.97 g/mol + 64.00 g/mol = 163.94 g/mol
By following these steps, you accurately calculate the molar mass of sodium phosphate. This calculation isn't just an academic exercise; understanding the molar mass of compounds like sodium phosphate is fundamental for various applications in chemistry and beyond.
Practical Example: Using the Molar Mass in a Real-World Context
Let’s consider a practical scenario where knowing the molar mass of sodium phosphate is essential. Suppose you’re working on a project that requires synthesizing a specific amount of sodium phosphate for a biochemical experiment. You need to prepare 50 grams of Na₃PO₄. To find out how many moles of sodium phosphate you’ll be working with, you divide the total mass by the molar mass.
Using the molar mass we calculated: 163.94 g/mol, the calculation is as follows:
Moles of Na₃PO₄ = 50 grams / 163.94 g/mol = 0.305 moles
This example demonstrates the importance of knowing the molar mass in practical applications, such as ensuring the correct quantities are used in experiments.
Practical FAQ
What if I need to calculate the molar mass for a different sodium phosphate compound?
If you’re working with a different sodium phosphate compound, the approach remains the same, but you’ll need to adjust the formula and the quantities of each element accordingly. Always start by writing down the chemical formula and then follow the step-by-step guide using the updated quantities and atomic masses. For example, for sodium dihydrogen phosphate (NaH₂PO₄), you would calculate the molar mass by considering the different number of sodium, phosphorus, hydrogen, and oxygen atoms present in the compound.
Understanding how to calculate the molar mass of sodium phosphate, and adapting the method for other compounds, is a valuable skill that enhances your analytical capabilities in chemistry and related fields. With these guidelines, you're well-equipped to tackle molar mass calculations with confidence and accuracy.
Remember, the key to mastering this process lies in breaking down the task into clear, manageable steps and applying practical examples to solidify your understanding. With practice, these calculations will become second nature, allowing you to focus more on the broader applications of your knowledge.