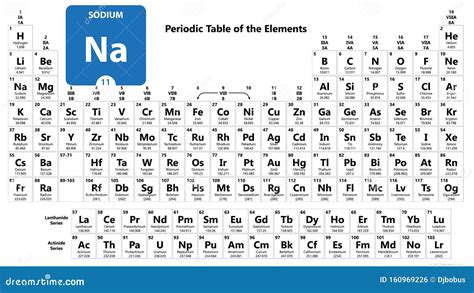
Welcome to our comprehensive guide on sodium, a critical element on the periodic table that holds immense importance in various fields, from chemistry to everyday life. This guide will provide you with detailed, practical insights on how to understand and utilize sodium effectively. Whether you're a student, a chemistry enthusiast, or someone simply curious about the elements that make up our world, this guide will address your needs with actionable advice and real-world examples.
Understanding Sodium: A Problem-Solution Approach
Sodium, symbolized by Na and possessing the atomic number 11, is a highly reactive, soft metal. Its importance spans numerous applications, from cooking salt to playing a crucial role in the human body as an electrolyte. However, the challenge often lies in understanding its reactive nature and safely handling it. This guide aims to demystify sodium, offering straightforward solutions to its inherent problems, such as managing its reactivity and ensuring its safe use in both chemical experiments and everyday tasks.
Why Sodium Matters:
Sodium is essential not only for chemical reactions but also for our everyday lives. In your body, it helps regulate blood pressure, nerve function, and fluid balance. Yet, there’s a fine line between beneficial and harmful sodium levels. Mismanagement can lead to problems, including chemical burns in labs or excess consumption leading to health issues in daily life.
Quick Reference: Key Sodium Insights
Quick Reference
- Immediate action item: When handling sodium metal, always keep it submerged in mineral oil to prevent reactive reactions with air and moisture.
- Essential tip: To convert sodium chloride (table salt) into sodium metal in a lab setting, use electrolysis, ensuring to follow safety protocols rigorously.
- Common mistake to avoid: Not recognizing that sodium compounds, like sodium hydroxide, can be highly corrosive. Always use protective gear when handling these substances.
Detailed How-To: Handling Sodium Metal
Sodium metal is a fascinating yet hazardous substance. This section delves into the precise methods for safely handling and utilizing sodium metal in both professional and educational settings.
Sodium metal, with its unique properties, can ignite upon exposure to air or water. Here's how to manage it safely:
Step-by-Step Guidance for Sodium Metal Handling
- Storage: Sodium must be stored in a sealed, moisture-proof container, preferably submerged in mineral oil to prevent any contact with air or water.
- Transfer: When transferring sodium, use appropriate containers designed to hold it in an oil bath. Always avoid plastic as it can react with sodium.
- Reaction Control: In the lab, when a reaction with sodium is necessary, ensure that you’re in a well-ventilated area, preferably under a fume hood, with safety equipment like gloves and goggles.
Real-World Application
In industries such as petroleum refining, sodium is used to remove sulfur from fuel gas streams. To implement this process:
- Utilize a sodium sponge process in which sodium vapor reacts with sulfur, forming sodium sulfide, which can be easily separated.
- Ensure that the process is conducted under controlled temperatures and pressures, minimizing any risks associated with sodium's reactive nature.
Detailed How-To: Using Sodium Compounds
Sodium compounds are widely used in various applications, from cleaning agents to medical treatments. Here’s how to work with these compounds effectively and safely.
Step-by-Step Guide for Sodium Compounds
- Sodium Hydroxide: Used extensively in cleaning, this compound can be corrosive. When handling it:
- Always wear protective gloves, goggles, and clothing to prevent skin and eye contact.
- Store it in a sealed container away from moisture and incompatible substances.
- Sodium Chloride: The common table salt, with critical health and culinary uses:
- In medical applications, it’s used in intravenous fluids and nasal sprays. When preparing solutions:
- Use sterile equipment and follow dosage instructions to ensure safety and effectiveness.
- In cooking, it’s crucial to use it judiciously due to health concerns associated with high sodium consumption.
Best Practices
To optimize the use of sodium compounds:
- For cleaning agents containing sodium hydroxide, perform dilutions in a well-ventilated area, always adding the compound to water, not the reverse, to avoid exothermic reactions.
- When using sodium chloride for medical or culinary purposes, adhere strictly to prescribed or recommended dosages and storage conditions.
Practical FAQ: Addressing Common Questions
What are the safety precautions for using sodium compounds in a lab?
Safety with sodium compounds is paramount. Here’s what you should follow:
- Always wear appropriate personal protective equipment (PPE) including gloves, goggles, and lab coats.
- Ensure that your workspace is clean and free of any materials that could react with sodium compounds.
- Store sodium compounds in their designated containers, away from moisture, heat, and incompatible substances.
- In case of a spill or exposure, know the immediate steps to take, such as rinsing with water for skin exposure and having emergency contact information readily available.
This guide aims to equip you with the knowledge and tools needed to understand and utilize sodium effectively, ensuring safety and efficiency in all applications. By adhering to these guidelines, you can navigate the complexities of sodium with confidence and proficiency.