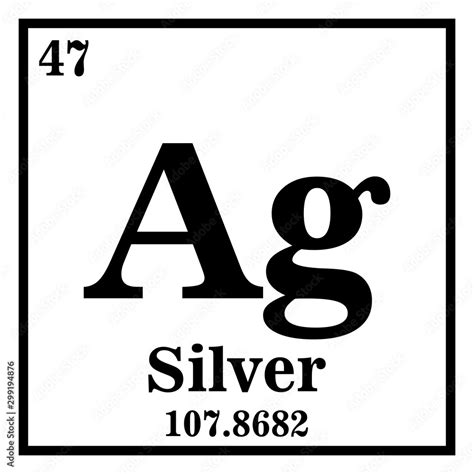Understanding the Power of Silver's Secret Spot on the Periodic Table: A Practical Guide
Are you curious about the unique properties of silver and how it's positioned on the periodic table? Many people know silver as a precious metal, but few understand the science behind its remarkable placement and what this means for its applications in various fields. This guide will demystify silver's secret spot on the periodic table, focusing on practical advice, real-world examples, and actionable tips that you can apply immediately.
Silver stands out as a versatile and valuable element. With its high conductivity, reflectivity, and catalytic properties, it’s essential in industries ranging from electronics to medicine. Understanding its positioning on the periodic table not only helps in appreciating its applications but also aids in making informed decisions when working with silver or considering its use in your projects.
Quick Reference
Quick Reference
- Immediate action item: Identify the atomic number and symbol of silver (Ag, 47) to understand its chemical properties.
- Essential tip: Silver is found in Group 11 and Period 5. This placement influences its high conductivity and lustrous appearance.
- Common mistake to avoid: Confusing silver with other transition metals like copper or gold; recognize silver’s distinct position for appropriate applications.
Detailed How-To Sections
Exploring Silver's Position on the Periodic Table
Understanding where silver is located on the periodic table gives you a head start in leveraging its unique properties. Let’s dive deeper into its position:
Silver (Ag) has an atomic number of 47 and is classified as a transition metal. On the periodic table, it’s found in Group 11, which signifies its place in the d-block, specifically in Period 5.
Group 11 metals share common traits such as high electrical and thermal conductivity, malleability, and luster. Silver, in particular, excels in these aspects due to its high electronegativity and ability to efficiently conduct both heat and electricity. These characteristics make it indispensable in high-tech industries.
Step-by-Step Guide to Silver’s Applications
Now that we’ve pinpointed silver’s spot, let’s go through its practical applications, step-by-step:
Electrical Conductivity
Silver’s unparalleled conductivity makes it the material of choice for various electrical applications:
- Step 1: Identify electrical components where superior conductivity is essential, such as in advanced electronics, photovoltaics, and high-speed circuits.
- Step 2: Compare the conductivity of other metals (like copper) with silver. Note that silver has about 60% better conductivity than copper, which makes it more efficient for high-demand applications.
- Step 3: When working on designs or projects, select materials accordingly to benefit from silver’s superior conductivity, thereby enhancing the performance and efficiency of the components.
Industrial Catalysis
Silver’s catalytic properties make it crucial in several chemical processes:
- Step 1: Recognize processes where silver can act as a catalyst. This includes water purification, gas production, and organic synthesis.
- Step 2: Research specific reactions where silver enhances reaction rates or selectivity. For instance, in ethylene oxide production, silver acts as a catalyst to improve efficiency and reduce costs.
- Step 3: Implement silver catalysts in industrial settings to benefit from its efficiency and effectiveness, leading to better product quality and economic benefits.
Medical and Healthcare Uses
Silver has long been valued for its antimicrobial properties:
- Step 1: Identify products where antimicrobial properties are crucial, such as wound dressings, medical devices, and hospital sanitation products.
- Step 2: Evaluate the benefits of silver nanoparticles in medical applications. For example, silver nanoparticles are embedded in dressings to prevent infections in burns and wounds.
- Step 3: Collaborate with healthcare professionals to integrate silver-based products, thereby ensuring better patient outcomes and reducing infection rates.
Practical FAQ Section
Why is silver so highly valued in the electronics industry?
Silver’s high electrical conductivity and excellent thermal conductivity make it an ideal choice for high-performance electronic components. It ensures efficient power transfer and heat dissipation, which are critical for the functioning of advanced electronic devices such as computers, smartphones, and medical equipment. Moreover, silver’s resistance to oxidation and its durability make it a preferred material for these applications.
How does silver’s positioning on the periodic table influence its antimicrobial properties?
Silver’s placement in Group 11 and Period 5 of the periodic table gives it unique electronic properties, which facilitate its antimicrobial action. The element’s ability to disrupt bacterial DNA and membranes, leading to cell death, is enhanced by its chemical stability and the formation of silver ions. These ions interact with proteins and enzymes in bacteria, preventing their replication and thus offering effective antimicrobial benefits.
By following this guide, you can better understand silver's unique position on the periodic table and how to leverage its properties in practical applications. Whether you’re an engineer, a scientist, or just a curious individual, this guide provides you with the essential knowledge to unlock the full potential of silver.
Remember, silver’s versatility is rooted in its precise chemical and physical properties, so take advantage of its unique spot on the periodic table to innovate and enhance your projects with this remarkable element.
If you have more questions or need further insights, continue exploring the applications and research surrounding silver to find new and exciting ways to utilize this valuable element in your field of work or interest.