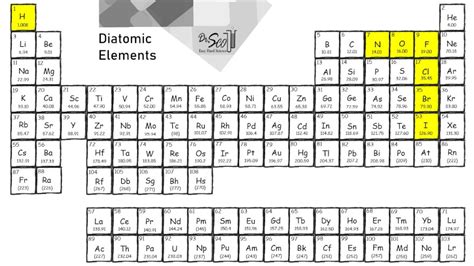
Seven diatomic elements, comprising only two atoms of a single element, are fundamental to the world of chemistry. This unique structural simplicity helps in understanding various chemical and physical properties, making these elements pivotal in both theoretical and practical applications. Hydrogen, nitrogen, oxygen, fluorine, chlorine, bromine, and iodine are the stars of this group.
Key Insights
- Understanding the reactivity and bonding in diatomic molecules enhances chemical process efficiency
- These elements play a critical role in synthesis and environmental studies
- Diatomic elements offer an excellent foundation for teaching fundamental chemistry concepts
The Chemistry Behind Diatomic Elements
Diatomic molecules represent a core area of study in chemistry. Their unique simplicity allows for straightforward analysis of molecular bonds and interactions. Each diatomic element exhibits distinct reactivity due to differing atomic structures and energies. For instance, molecular oxygen (O₂) demonstrates significant reactivity, driving combustion processes. Conversely, nitrogen (N₂) is relatively inert under standard conditions, making it an essential component of our atmosphere and inert environments in laboratory settings.Applications in Industrial and Environmental Chemistry
Diatomic elements hold immense importance in industrial processes and environmental chemistry. For example, chlorine (Cl₂) is a crucial disinfectant and is widely used in water treatment. Additionally, in the industrial realm, chlorine is pivotal in producing various chemicals such as PVC (polyvinyl chloride). Bromine (Br₂), often used in flame retardants, underscores the utility of these elements in safety and commercial applications. Their environmental impact, however, necessitates careful regulation to mitigate adverse ecological effects.What makes diatomic elements unique?
Diatomic elements consist of only two atoms of the same element, allowing for a simplified understanding of molecular interactions and chemical bonding.
How do diatomic elements influence environmental studies?
Diatomic elements such as chlorine and bromine play a significant role in pollution studies and regulatory measures due to their impact on air and water quality. Understanding their behavior helps in developing strategies to control environmental hazards.
The seven diatomic elements embody both the elegance and complexity of molecular chemistry. Their applications in various fields underscore the necessity of a deep understanding of their properties and behaviors. Through rigorous study and practical application, we unlock nature’s chemistry secrets, enabling advancements that benefit both scientific knowledge and everyday life.