Welcome to our comprehensive guide on Potassium Hydroxide (KOH) molar mass. This guide is designed to walk you through everything you need to know about calculating and understanding KOH’s molar mass, breaking down complex concepts into simple, actionable advice. Whether you’re a student, a professional chemist, or simply curious about this essential chemical compound, this guide will provide you with the knowledge and practical tools you need.
Understanding the Problem: Why Molar Mass Matters
Calculating the molar mass of Potassium Hydroxide is crucial in chemistry because it provides a fundamental understanding of the substance’s composition and properties. Molar mass, expressed in grams per mole (g/mol), is vital for solving stoichiometric problems, determining the amount of reactants needed for a chemical reaction, and understanding the material properties. If you’re dealing with KOH for laboratory experiments, industrial applications, or academic studies, mastering its molar mass calculation can significantly improve your accuracy and efficiency.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Always check the latest atomic weights from the periodic table for the most accurate calculations.
- Essential tip with step-by-step guidance: Start by determining the molar masses of each element in KOH, then add them together.
- Common mistake to avoid with solution: Confusing molecular and empirical formulas. Ensure you’re calculating the molar mass for KOH, not any variation like KOH₂.
How to Calculate the Molar Mass of KOH
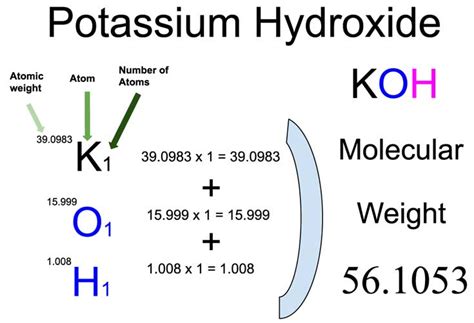
Calculating the molar mass of Potassium Hydroxide involves summing the atomic masses of its constituent elements: potassium (K), oxygen (O), and hydrogen (H). Here’s how to do it step-by-step:
-
Step 1: Find Atomic Weights:
Locate the atomic weights of each element in the periodic table. For accurate results, use the most up-to-date weights. For KOH, these are approximately:
- Potassium (K): 39.10 g/mol
- Oxygen (O): 16.00 g/mol
- Hydrogen (H): 1.01 g/mol
-
Step 2: Sum the Atomic Weights:
Add the atomic weights of all the elements in the formula. KOH has one potassium atom, one oxygen atom, and one hydrogen atom, so:
Molar mass of KOH = (1 × 39.10 g/mol) + (1 × 16.00 g/mol) + (1 × 1.01 g/mol) = 39.10 g/mol + 16.00 g/mol + 1.01 g/mol = 56.11 g/mol
-
Step 3: Verify Your Calculation:
It’s always good practice to double-check your calculations. Make sure you’ve correctly identified the atomic weights and that you’ve added them in the right proportions.
Practical Examples and Applications
To ensure that you fully grasp the concept, let’s look at some practical examples of how to apply the molar mass of KOH in various scenarios:
Example 1: Preparing a KOH Solution
Suppose you need to prepare a 0.5 M KOH solution in a 1-liter flask for a laboratory experiment. To determine the amount of KOH you need, follow these steps:
-
Step 1: Calculate the moles of KOH required:
To prepare a 0.5 M solution in 1 liter, you need 0.5 moles of KOH. Since the molar mass of KOH is 56.11 g/mol, you need:
0.5 moles × 56.11 g/mol = 28.055 grams of KOH
-
Step 2: Weigh out the KOH:
Use an analytical balance to accurately weigh 28.055 grams of KOH.
-
Step 3: Dissolve the KOH:
Slowly add the KOH to a flask containing approximately 800 ml of distilled water. Stir until the KOH is completely dissolved, then add more water to bring the total volume to 1 liter.
Example 2: Stoichiometric Calculations
In a reaction where KOH neutralizes hydrochloric acid (HCl), understanding the molar mass is essential for accurate stoichiometric calculations. For instance:
Let’s determine how many grams of KOH are needed to neutralize 500 ml of 0.25 M HCl solution.
-
Step 1: Calculate moles of HCl:
To find the moles of HCl, use the concentration and volume:
0.25 moles/liter × 0.5 liters = 0.125 moles of HCl
-
Step 2: Use the stoichiometry of the reaction:
The balanced chemical equation is:
KOH + HCl → KCl + H₂O
From the equation, one mole of KOH neutralizes one mole of HCl, so you need 0.125 moles of KOH.
-
Step 3: Calculate the mass of KOH needed:
To find the mass of KOH, multiply the moles by the molar mass:
0.125 moles × 56.11 g/mol = 6.014 grams of KOH
Practical FAQ
How do I determine if KOH is in excess in my reaction?
To determine if KOH is in excess, you need to complete the reaction stoichiometry calculations. Start by finding out how much KOH is required to neutralize your acid (HCl in this case) based on its concentration and volume. If you have more KOH than the calculated amount, it’s in excess. For example, if you need 6.014 grams to react completely with 500 ml of 0.25 M HCl and you used 7 grams, you have 0.986 grams in excess.
What safety precautions should I take when handling KOH?
Potassium hydroxide is caustic and can cause severe burns upon contact with skin or eyes. Always wear appropriate personal protective equipment (PPE) such as gloves, goggles, and lab coat. Work in a well-ventilated area or under a fume hood, and store KOH in tightly closed containers away from moisture and incompatible substances like acids or organic materials.
Best Practices and Tips
Following these best practices will help you ensure accurate calculations and safe handling of Potassium Hydroxide:
-
Double-check your calculations:
Always verify the molar mass and stoichiometric calculations to avoid errors.
-
Use accurate measuring tools:
Employ analytical balances for precise weighing and graduated cylinders for accurate volume measurements.
- <