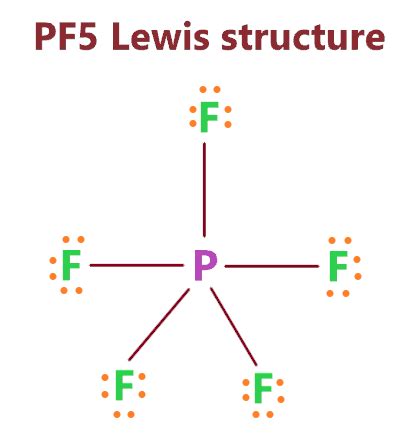
Understanding the PF5 Lewis structure is a pivotal aspect of grasping molecular geometry and its influence on chemical properties. This comprehensive guide aims to decode the PF5 molecule’s Lewis structure, unveiling the secrets behind its shape and the implications for its reactivity. Through this journey, you will gain step-by-step insights, practical examples, and actionable tips to make this complex subject approachable and understandable.
Problem-Solution Opening Addressing User Needs
Many chemistry students encounter challenges when trying to determine the molecular geometry of complex molecules like phosphorus pentafluoride (PF5). The PF5 molecule is an ideal case study to understand the relationship between a molecule’s Lewis structure and its three-dimensional geometry. Often, the process of drawing the Lewis structure, predicting its geometry, and understanding the underlying principles can seem daunting. This guide aims to demystify the PF5 Lewis structure by breaking down each step with detailed instructions and practical examples, ensuring that even those new to the subject can grasp the concepts with confidence.
By the end of this guide, you will not only be able to draw the PF5 Lewis structure accurately but also comprehend why the molecule adopts its specific geometry. This knowledge is not just academic; understanding molecular geometry has practical applications in fields such as pharmaceuticals, materials science, and environmental chemistry.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Start by drawing the skeleton structure of the molecule, placing the phosphorus atom in the center and surrounding it with five fluorine atoms.
- Essential tip with step-by-step guidance: Ensure that phosphorus uses its 3d orbitals for bonding, allowing it to form five covalent bonds with fluorine atoms, fulfilling the octet rule.
- Common mistake to avoid with solution: Avoid the mistake of believing that the central atom must always satisfy the octet rule by itself; in PF5, phosphorus uses 3d orbitals, which expands its bonding capacity beyond the traditional octet rule.
Detailed How-To: Drawing the PF5 Lewis Structure
Let’s delve into the detailed process of drawing the PF5 Lewis structure step-by-step. By following these instructions, you will gain a clear understanding of the structure and its geometry.
The first step is to identify the total number of valence electrons. Phosphorus (P) is in group 15 and has 5 valence electrons, while each fluorine (F) atom, in group 17, has 7 valence electrons. Given there are five fluorine atoms, you calculate the total valence electrons as follows:
Total valence electrons = 5 (from P) + 5 * 7 (from F) = 5 + 35 = 40 valence electrons
Next, arrange the atoms in the molecule's skeleton structure. Phosphorus is the central atom, surrounded by five fluorine atoms. The structure looks like this:
P-F-F-F-F-F
To form the bonds, start placing two electrons (a single bond) between the central phosphorus atom and each of the five fluorine atoms. This accounts for 10 electrons:
P: :F:—F:—F:—F:—F:
Now, distribute the remaining electrons. With a total of 40 valence electrons and having already placed 10 in the bonds, you have 30 electrons left to place. The fluorine atoms each need 8 electrons to fulfill the octet rule. The remaining electrons will be added to the central phosphorus atom to complete its octet (8 electrons) as well.
Since phosphorus uses its 3d orbitals to expand its bonding capacity, it can hold more than 8 electrons. Place the remaining electrons in pairs around the phosphorus atom:
P: :F:—F:—F:—F:—F:
Now, you have a complete Lewis structure with no lone pairs on the fluorine atoms and an expanded octet on the phosphorus atom.
With this structure in hand, we can predict the molecular geometry. PF5 has a total of six electron pairs (five bonding pairs and one lone pair on phosphorus). According to the VSEPR (Valence Shell Electron Pair Repulsion) theory, these pairs arrange themselves to minimize repulsion, resulting in a trigonal bipyramidal geometry.
Detailed How-To: Predicting the Molecular Geometry of PF5
Understanding the geometry of a molecule like PF5 is crucial for predicting its chemical behavior and physical properties. Here, we break down the process of predicting the geometry of the PF5 molecule using the VSEPR theory.
Step 1: Count the electron pairs around the central atom. In PF5, the central phosphorus atom is surrounded by five bonding pairs from the fluorine atoms and one lone pair. This gives us a total of six electron pairs.
Step 2: Determine the electron-pair geometry using the VSEPR theory. For six electron pairs, the geometry is described as an octahedral arrangement. However, since we only have five fluorine atoms as ligands, the geometry reduces to a trigonal bipyramidal shape.
Step 3: Identify the molecular geometry. For PF5, the five fluorine atoms occupy equatorial and axial positions in a trigonal bipyramidal arrangement. The axial positions have a linear arrangement, while the equatorial positions form a trigonal planar shape. All fluorine atoms are equivalent, and there are no lone pairs on the fluorine atoms, resulting in a perfect trigonal bipyramidal molecular geometry.
This understanding helps in predicting the molecule's reactivity and its interactions in chemical reactions.
Practical FAQ
Why does phosphorus use its 3d orbitals in PF5?
Phosphorus, despite being in the third period, can utilize its 3d orbitals for bonding due to its position in the periodic table. The expansion of the octet beyond eight electrons by using 3d orbitals allows phosphorus to form five bonds with fluorine atoms in PF5. This is an exception to the general octet rule for second-period elements, which typically use only their 2s and 2p orbitals.
Can PF5 have different geometries based on lone pairs?
PF5 does not have lone pairs on the fluorine atoms. However, if there were any, they would influence the molecular geometry. For instance, if an additional lone pair were present on phosphorus, it would occupy an equatorial position, potentially changing the geometry slightly from trigonal bipyramidal to a different arrangement like see-saw or T-shape, depending on the number of lone pairs.
How does the molecular geometry of PF5 affect its chemical properties?
The trigonal bipyramidal geometry of PF5 affects its reactivity and polarity. With no net dipole moment, PF5 is a nonpolar molecule. Its symmetrical structure makes it less reactive compared to molecules with more polar or asymmetrical geometries. Additionally, the spatial arrangement of fluorine atoms can influence how it interacts with other molecules in chemical reactions.
This guide has covered the essentials of decoding the PF5 Lewis structure and understanding its molecular geometry. By understanding the step-by-step process of drawing the Lewis structure and predicting the molecular geometry, you now have the tools to apply these principles to other molecules. Remember, practice is key to mastering these concepts, so continue to explore and apply these techniques to different molecules to deepen your understanding.