Phosphorus trichloride (PCl3) is an essential compound in the field of organic chemistry and materials science, often acting as an intermediate in the synthesis of various chemicals. Understanding the molecular geometry of PCl3 provides valuable insights into its reactivity, properties, and applications. In this article, we will delve into the secrets of PCl3’s molecular geometry, providing a comprehensive analysis based on expert perspectives and practical insights.
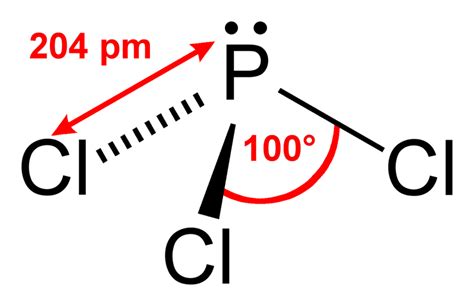
The molecular geometry of PCl3 can be deciphered using the VSEPR (Valence Shell Electron Pair Repulsion) theory. This theory allows us to predict the shape of molecules by considering how electron pairs around a central atom will arrange themselves to minimize repulsion. PCl3 consists of a central phosphorus atom bonded to three chlorine atoms. The phosphorus atom also has one lone pair of electrons, contributing to the overall electron pair count around the phosphorus.
Key Insights
- Primary insight with practical relevance: The molecular geometry of PCl3 is trigonal pyramidal, which greatly influences its reactivity and interaction with other molecules.
- Technical consideration with clear application: Understanding the polarity of PCl3 is essential for predicting its solubility and interactions in various chemical environments.
- Actionable recommendation: Utilizing molecular modeling software can provide a visual understanding of the PCl3 geometry, further aiding in the interpretation of its chemical behavior.
Trigonal Pyramidal Geometry of PCl3
PCl3 exhibits a trigonal pyramidal geometry. According to VSEPR theory, the phosphorus atom in PCl3 is sp3 hybridized, meaning one s orbital and three p orbitals are mixed to form four sp3 hybrid orbitals. Three of these hybrid orbitals overlap with the 1s orbitals of the three chlorine atoms, while the fourth contains the lone pair of electrons. This arrangement results in a trigonal pyramidal shape, with bond angles slightly less than 109.5 degrees, due to the repulsion exerted by the lone pair.
Polarity and Dipole Moment
The molecular geometry of PCl3 leads to its polarity. Due to the lone pair on the phosphorus atom, there is an asymmetry in the electron distribution, resulting in a net dipole moment. The electronegativity difference between phosphorus and chlorine also contributes to the polarity. As a result, PCl3 is a polar molecule, which affects its solubility in polar solvents and its reactivity with other molecules.
Does PCl3 have a net dipole moment?
Yes, PCl3 has a net dipole moment due to its trigonal pyramidal geometry and the presence of a lone pair on the phosphorus atom, which creates an asymmetry in electron distribution.
What is the application of PCl3 in organic synthesis?
PCl3 is commonly used in organic synthesis to convert alcohols into chlorides. It acts as a chlorinating agent, facilitating the substitution of hydroxyl groups with chlorine atoms.
To conclude, understanding the molecular geometry of PCl3 is crucial for comprehending its chemical behavior. The trigonal pyramidal shape, arising from sp3 hybridization and lone pair repulsion, along with its polarity, plays a significant role in its interactions and applications in organic chemistry. Utilizing these insights can significantly enhance the practical application and understanding of PCl3 in various scientific domains.