Understanding the significance of particle per million (ppm) in environmental testing is critical for accurate and effective monitoring of pollutants and contaminants in various ecosystems. This level of precision facilitates the detection of even minute quantities of harmful substances, which is vital for ensuring public health and environmental safety.
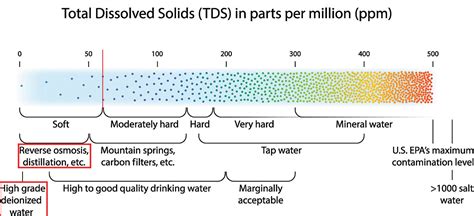
PPM, or parts per million, is a standard measure of concentration used extensively in environmental science to express the ratio of a substance to the total mixture of materials it is a part of. Specifically, it represents the number of units of a particular substance per million units of the total mixture. This level of specificity is paramount in detecting and measuring pollutants at very low concentrations, often found in air, water, soil, or other environmental matrices.
Here are the primary insights around the understanding of ppm in environmental testing:
Key Insights
- PPM provides precision in detecting trace pollutants, essential for maintaining environmental safety.
- Analytical techniques like gas chromatography and mass spectrometry play a crucial role in accurate ppm measurements.
- Adhering to regulatory standards through precise ppm assessments ensures compliance and mitigates environmental risks.
Environmental testing that employs ppm is vital for monitoring pollution in air, water, and soil. For example, in air quality monitoring, ppm is used to measure pollutants like volatile organic compounds (VOCs), nitrogen dioxide (NO2), sulfur dioxide (SO2), and carbon monoxide (CO). These measurements are essential for assessing health risks to populations living in affected areas.
Application in Air Quality Monitoring
Air quality testing that utilizes ppm is fundamental for identifying and regulating harmful pollutants. For instance, industries often release VOCs, which can lead to both immediate health hazards and long-term environmental damage. Detecting these VOCs at ppm levels allows environmental agencies to enforce regulations that limit emissions and protect public health.
Usage in Water and Soil Quality Assessment
Water and soil quality analysis also benefits significantly from ppm measurements. Contaminants like heavy metals (e.g., lead, mercury) and pesticides must be quantified at ppm levels to evaluate their impact on ecosystems and human health. Advanced techniques like inductively coupled plasma mass spectrometry (ICP-MS) allow scientists to detect these contaminants accurately, facilitating better-informed decisions on remediation strategies and regulatory compliance.
Why is ppm important in environmental testing?
PPM is crucial for detecting minute concentrations of pollutants in the environment. It provides the precision necessary for regulatory compliance, public health protection, and environmental conservation efforts.
What techniques are commonly used to measure ppm levels?
Techniques like gas chromatography (GC), mass spectrometry (MS), and inductively coupled plasma mass spectrometry (ICP-MS) are commonly used for measuring ppm levels in environmental samples due to their high sensitivity and accuracy.
In conclusion, understanding and applying the concept of particle per million in environmental testing is vital for ensuring the highest level of scrutiny in detecting and managing pollutants. The ability to measure and interpret these low levels of contaminants allows for stringent regulatory oversight, informed remediation efforts, and ultimately, the protection of our health and environment.