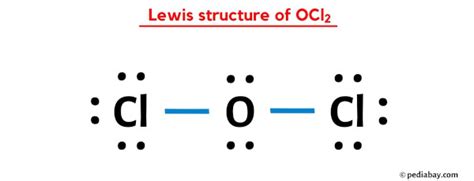
OCL2, also known as dichlorine oxide, can be a tricky molecule to decipher if you’re just starting out with Lewis structures. Many students struggle to correctly represent its electron distribution, leading to confusion about formal charges, resonance, and stability. This guide aims to unravel the mysteries of the OCL2 Lewis structure, providing you with step-by-step instructions, practical examples, and clear solutions to common problems. Let’s dive in and decode this compound together!
Understanding the Basics of OCL2 Lewis Structure
Before we jump into the intricacies, it’s crucial to understand the fundamental principles of Lewis structures. The Lewis structure of a molecule helps us visualize the valence electrons and how they form bonds between atoms. For OCL2, the goal is to place the least electronegative atom in the center (oxygen), surround it with more electronegative atoms (chlorine in this case), and follow the octet rule to ensure that each atom has a stable electron configuration.
Problem-Solution Opening Addressing User Needs
If you’ve found yourself perplexed by the OCL2 Lewis structure, you’re not alone. Many students often grapple with how to correctly distribute the electrons and understand the formal charge of each atom. This guide is designed to give you clear, practical, and actionable advice to demystify the process. Whether you’re learning for the first time or revisiting to refresh your knowledge, we will walk through each step methodically to ensure you can confidently create the OCL2 Lewis structure. We will also address common pitfalls and explain practical examples to help reinforce your understanding.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Draw the skeletal structure of OCL2 first by placing the central atom (oxygen) and connect it with chlorines.
- Essential tip with step-by-step guidance: Distribute electrons starting from the least electronegative atom to ensure proper bond formation.
- Common mistake to avoid with solution: Not checking formal charges; ensure that the sum of formal charges equals the charge of the molecule.
How to Draw the OCL2 Lewis Structure: Step-by-Step Guide
Let’s break down the process of drawing the OCL2 Lewis structure.
Step 1: Calculate Total Valence Electrons
Start by determining the total number of valence electrons available for the OCL2 molecule. Oxygen has 6 valence electrons, and each chlorine has 7. Hence:
- Valence electrons of Oxygen: 6
- Valence electrons of Chlorine: 2 (since there are two chlorines) × 7 = 14
- Total valence electrons: 6 + 14 = 20
Step 2: Draw the Skeletal Structure
Begin by sketching the skeletal structure of the molecule, where the central atom (oxygen) is bonded to the two surrounding chlorines:
O—Cl—Cl
Step 3: Distribute Electrons to Form Bonds
Distribute the electrons to form single bonds between the central atom (oxygen) and each chlorine atom:
O—Cl—Cl
Step 4: Complete the Octet Rule for Outer Atoms
Each chlorine atom now has an octet (8 electrons). However, oxygen does not have a complete octet with only two single bonds (it has 4 electrons).
Step 5: Place Remaining Electrons on Central Atom
Distribute the remaining electrons to complete the octet around the central atom. For OCL2, you will place 6 more electrons (3 lone pairs) around the oxygen atom:
:O—Cl—Cl:
Step 6: Check Formal Charges
To ensure your structure is correct, calculate the formal charges for each atom:
- For Oxygen:
- Valence electrons = 6
- Non-bonding electrons = 6 (3 lone pairs)
- Bonding electrons = 4 (2 bonds)
- Formal charge = 6 - 6 - 4/2 = 0
- For Chlorine:
- Valence electrons = 7
- Non-bonding electrons = 6 (3 lone pairs)
- Bonding electrons = 2 (1 bond)
- Formal charge = 7 - 6 - 2/2 = 0
Step 7: Confirm the Resonance Structures
Although the given structure for OCL2 does not require resonance, it’s crucial to understand that some molecules might have more than one valid Lewis structure due to resonance. However, OCL2 does not exhibit resonance structures.
Practical Example of OCL2 Lewis Structure
Let’s visualize this process with a practical example:
Consider an advanced problem where you need to draw the OCL2 Lewis structure and ensure it’s balanced. Follow the steps:
- Calculate total valence electrons: 20
- Draw skeletal structure: O—Cl—Cl
- Distribute electrons to form bonds: O—Cl—Cl
- Complete the octet for outer atoms (chlorines already have complete octets)
- Place 6 remaining electrons around oxygen (3 lone pairs)
- Calculate formal charges to confirm neutrality (0 formal charge for each atom)
Practical FAQ
What is the most common mistake when drawing the OCL2 Lewis structure?
The most common mistake is neglecting to calculate formal charges. It’s crucial to ensure that the sum of formal charges equals the overall charge of the molecule. For OCL2, double-check the formal charges of both oxygen and chlorine atoms to confirm correctness.
Can the OCL2 molecule exhibit resonance structures?
No, the OCL2 molecule does not exhibit resonance structures. It has a stable Lewis structure where all atoms achieve their octet rule satisfaction without the need for multiple forms.
How can I ensure that my Lewis structure is correct?
To ensure your Lewis structure is correct, follow these steps:
- Calculate total valence electrons
- Place the central atom and connect with outer atoms
- Distribute electrons to form bonds
- Complete the octet for outer atoms
- Place remaining electrons on the central atom to complete its octet
- Calculate formal charges and ensure they sum up to zero for a neutral molecule
This comprehensive guide should help you master the OCL2 Lewis structure with clear, practical examples and step-by-step instructions. Remember, practice makes perfect, so keep practicing drawing different Lewis structures to solidify your understanding.