Understanding the molecular shape of nitrite (NO3) is fundamental to grasping advanced concepts in chemistry, especially when delving into the realms of molecular geometry, resonance structures, and chemical bonding. The shape of the nitrite ion plays a pivotal role in predicting its chemical behavior, reactivity, and interactions with other molecules. In this article, we will explore the structural nuances of the NO3 molecular shape with an expert perspective, providing evidence-based statements and practical insights.
Key Insights
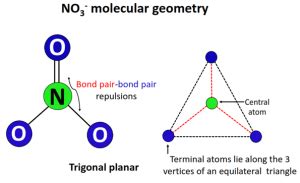
- The nitrite ion (NO3) adopts a trigonal planar molecular geometry.
- Resonance structures significantly affect the molecular shape and bond lengths.
- Understanding the NO3 shape aids in predicting the ion’s chemical behavior and reactivity.
Structural Analysis of NO3
The nitrite ion (NO3-) consists of one nitrogen atom centrally bonded to three oxygen atoms. According to the VSEPR (Valence Shell Electron Pair Repulsion) theory, the arrangement of electron pairs around the central atom influences the molecular geometry. In NO3-, the nitrogen atom is sp2 hybridized, resulting in a trigonal planar shape. This means that the nitrogen atom forms three sigma bonds with the oxygen atoms, and the shape minimizes the repulsion between these electron pairs, aligning them 120 degrees apart.
Resonance and Bond Lengths
An important aspect of NO3- is the concept of resonance. In reality, the nitrite ion does not have a single fixed structure. Instead, it exhibits resonance, where the electron distribution is shared among multiple valid structures. These resonance structures depict equivalent arrangements of double and single bonds between nitrogen and oxygen atoms. The actual structure of NO3- is a hybrid of these resonance forms, leading to the equal bond lengths in the ion. This uniform bond length is a consequence of delocalized electrons across the nitrogen and oxygen atoms, which is a key feature of resonance stabilization in the nitrite ion.
Chemical Behavior and Practical Implications
Understanding the NO3 molecular shape has practical implications in various chemical processes. For example, the trigonal planar geometry of NO3- allows it to interact differently compared to molecules with different geometries. This particular shape affects how NO3- engages in ionic interactions, its reactivity in acid-base reactions, and its overall stability in different chemical environments. This knowledge becomes particularly important when studying environmental chemistry, where nitrite ions play a role in processes like water treatment and soil chemistry.
Does NO3 exhibit stereoisomerism?
NO3- does not exhibit stereoisomerism because it has a symmetrical trigonal planar shape. Stereoisomers occur in molecules with different spatial arrangements of atoms, but NO3’s geometry does not allow for such variations.
How does the NO3 shape influence its reactivity?
The trigonal planar shape of NO3- facilitates specific types of chemical reactions, including its role as a reducing agent in various redox reactions. The equal bond lengths due to resonance stabilize the ion, making it reactive towards electron-deficient species.
In conclusion, deciphering the molecular shape of the nitrite ion (NO3-) provides significant insights into its chemical nature and behavior. The trigonal planar geometry, resonance stabilization, and implications in practical applications form the cornerstone of understanding this essential aspect of inorganic chemistry.