Problem-Solution Opening Addressing User Needs
Navigating the complexities of the International Union of Pure and Applied Chemistry (IUPAC) nomenclature for organic compounds can feel like trying to solve a puzzle with no manual. This seemingly intricate task is crucial for chemists and students alike to communicate precisely and avoid confusion in scientific writing and discussions. However, with a structured approach and the right guidance, mastering IUPAC nomenclature is within your grasp. This guide will break down the process into clear, manageable steps, offering practical tips and real-world examples to ensure you can name any organic compound correctly and confidently. Whether you are a seasoned chemist looking to refine your skills or a student embarking on your first foray into organic chemistry, this guide will equip you with the knowledge and confidence to name compounds according to IUPAC rules without stress or confusion.
Quick Reference
Quick Reference
- Immediate action item: Always start by identifying the longest continuous chain of carbon atoms in the compound (the parent chain) when you begin naming an organic compound.
- Essential tip: Follow the IUPAC naming conventions step-by-step: identify the parent chain, name substituents, and prioritize functional groups correctly.
- Common mistake to avoid: Overlooking the significance of the parent chain’s numbering, which determines the lowest possible locants for substituents and functional groups.
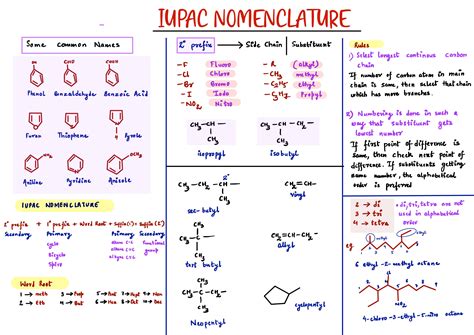
Understanding the Basics of IUPAC Nomenclature
Understanding IUPAC nomenclature starts with knowing the fundamental principles behind naming organic compounds. The key to this system is the parent chain, which is the longest continuous chain of carbon atoms in the molecule. This chain’s naming and numbering determine the base name of the compound and influence where substituents and functional groups are positioned. Let’s delve into these basics in detail:
Step 1: Identifying the Parent Chain
The parent chain is central to the IUPAC naming process. It’s the longest continuous chain of carbon atoms that determines the base name of the compound. For example, in a molecule with a choice between a four-carbon chain and a three-carbon chain, you would select the four-carbon chain as the parent.
Step 2: Naming the Parent Chain
Once you’ve identified the parent chain, you need to name it based on the number of carbon atoms it contains. The prefixes for the first few numbers are:
| Number of Carbons | Prefix |
|---|---|
| 1 | meth- |
| 2 | eth- |
| 3 | prop- |
| 4 | but- |
| 5 | pent- |
| 6 | hex- |
For example, a parent chain with four carbon atoms is named butane.
Step 3: Numbering the Parent Chain
Numbering the parent chain is crucial for assigning the lowest possible locants (numbers) to the substituents and functional groups. The goal is to assign the lowest set of locants to these elements. For instance, if a substituent can be numbered starting from either end of the chain, choose the end that gives the lowest number.
Detailed How-To: Naming Simple Alkanes
Let’s go through the process of naming simple alkanes, starting with straightforward examples and gradually introducing more complexity. This will help you build a solid foundation before tackling more complex compounds.
Example 1: Naming Propane
Propane is a simple alkane with a three-carbon parent chain. Here’s how to name it:
- Identify the longest chain: There’s only one chain of three carbons.
- Name the parent chain: According to the IUPAC prefixes, three carbons are named as ‘prop’.
- Determine the name: Add the suffix ‘-ane’ to indicate it’s an alkane, resulting in ‘propane’.
Example 2: Naming Butane
Butane has a four-carbon parent chain:
- Identify the longest chain: There’s a single chain of four carbons.
- Name the parent chain: Four carbons are named as ‘but’.
- Determine the name: Add the suffix ‘-ane’ to indicate it’s an alkane, resulting in ‘butane’.
Example 3: Naming with Substituents (2-methylpropane)
When dealing with substituents like methyl groups, the process slightly changes:
- Identify the longest chain: Find the longest continuous carbon chain. In this case, it’s a three-carbon chain.
- Name the parent chain: Three carbons are ‘prop’.
- Identify the substituent: There’s a methyl group (CH3) attached at the second carbon.
- Name the substituent: Methyl is named as ‘methyl’.
- Number the parent chain: Assign numbers starting from the end closest to the substituent. Here, the methyl group is on carbon 2.
- Combine the name: Use the locant ‘2-’ to denote the position of the methyl group, resulting in ‘2-methylpropane’.
Detailed How-To: Naming Compounds with Functional Groups
Functional groups add complexity to the names, and their correct prioritization is essential. Here, we’ll focus on naming compounds with common functional groups such as alcohols, ethers, and carbonyl compounds.
Example 1: Ethanol (an Alcohol)
Alcohols are named by replacing the ‘-ane’ suffix of the parent chain with ‘-anol’:
- Identify the longest chain: Determine the longest chain containing the hydroxyl (OH) group. This chain has two carbons.
- Name the parent chain: Two carbons are ‘eth’.
- Identify the functional group: The OH group indicates an alcohol.
- Name the functional group: Replace ‘-ane’ with ‘-anol’, resulting in ‘ethanol’.
Example 2: Ethyl Ether
Ethers have the general formula R-O-R’. The simplest ether is dimethyl ether:
- Identify the parent chains: Both chains are methyl (CH3).
- Name each parent chain: ‘Methyl’ for each chain.
- Combine the names: Use ‘oxy’ to indicate the oxygen linking the two substituents, resulting in ‘dimethyl ether’.
Example 3: Acetone (a Ketone)
Ketones are identified by replacing the ‘-ane’ suffix of the parent chain with ‘-one’:
- Identify the longest chain: The chain contains three carbons.
- Name the parent chain: Three carbons are ‘prop’.
- Identify the functional group: A carbonyl group (C=O) is present, making it a ketone.
- Name the functional group: Replace ‘-ane’ with ‘-one’, resulting in ‘propanone’.
- Identify the position of the carbonyl group: In acetone, it’s at carbon 2.
- Combine the name: No need to change the locant since it’s the default position for the carbonyl in a three-carbon chain, resulting in ‘acetone’.
Practical FAQ
How do I prioritize multiple functional groups in a compound?
Functional groups are prioritized based on their position in the IUPAC order of seniority. Here’s how to prioritize: