Understanding the NH3 (Ammonia) Lewis Structure is essential for grasping basic concepts in chemistry, especially in molecular bonding and structure. This guide will take you through a comprehensive and actionable approach to mastering the NH3 Lewis Structure, starting with an initial problem-solution opening, followed by a quick reference guide, detailed how-to sections, and practical FAQs.
If you're struggling with understanding the ammonia molecule's Lewis structure, you're not alone. The NH3 Lewis structure might seem daunting at first, but with the right approach and guidance, you'll find it easier to comprehend. This guide offers step-by-step advice, practical examples, and practical solutions to the common challenges faced by users like you. Let’s dive in and demystify NH3’s Lewis structure!
Quick Reference
Quick Reference
- Immediate action item: Draw a skeletal structure of NH3, placing the nitrogen atom in the center and hydrogen atoms around it.
- Essential tip: Always start by calculating the total number of valence electrons. For NH3, nitrogen has 5 and each hydrogen has 1, totaling 8 valence electrons.
- Common mistake to avoid: Forgetting to follow the octet rule; nitrogen is happy with 8 electrons, and each hydrogen gets its 2 electrons.
Let's break it down in a more detailed way.
Understanding the NH3 Lewis Structure: A Step-by-Step Guide
To master the NH3 Lewis structure, we need to follow a structured approach:
Step 1: Drawing the Skeleton
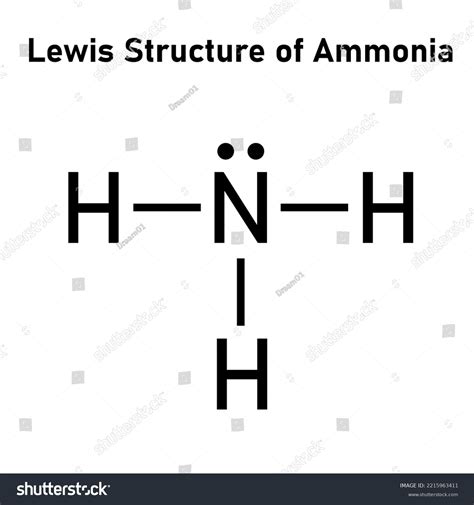
First, visualize NH3 and draw a basic skeleton. Place the nitrogen (N) atom in the center, as it is more electronegative and can accommodate the lone pairs better. Arrange three hydrogen (H) atoms around the nitrogen atom, each forming a single bond:
Structure:
H
|
H - N - H
|
H
Step 2: Calculate Total Valence Electrons
Next, tally up the valence electrons available. Nitrogen has 5, and there are three hydrogen atoms, each with 1 valence electron:
5 (from N) + 3*1 (from H) = 8 valence electrons
Step 3: Place Electrons Around the Atoms
Distribute these electrons to satisfy the octet rule (except for hydrogen, which only needs 2 electrons). Start by giving each hydrogen atom two electrons, forming single bonds:
Structure:
H
|
H - N - H
|
H
You’ve used 6 electrons (3 for each bond), so we have 2 electrons left. Place these remaining electrons on the nitrogen atom to satisfy its octet:
Step 4: Check for Octet Rule Compliance
Ensure nitrogen has an octet (8 electrons), and each hydrogen has 2 electrons:
Structure:
H
|
H - N - H
|
H
with lone pair represented on nitrogen:
H
|
H - N(:) - H
|
H
You’ve successfully drawn the NH3 Lewis structure!
Advanced Tips for Mastering the NH3 Lewis Structure
Beyond the basics, there are several advanced tips to refine your understanding:
Tip 1: Resonance Structures
Though not applicable for NH3, understanding the concept of resonance helps when dealing with molecules that can exhibit different Lewis structures due to delocalized electrons. Just keep it in mind for more complex molecules.
Tip 2: Formal Charge Calculation
Calculate the formal charge for each atom in your structure. Formal charge helps verify the stability of your Lewis structure. For NH3:
- Nitrogen: Valence electrons = 5, Non-bonding electrons = 2, Bonding electrons = 6 Formal Charge (N) = 5 - (2 + 6/2) = 0 - Hydrogen: Valence electrons = 1, Non-bonding electrons = 0, Bonding electrons = 2 Formal Charge (H) = 1 - (0 + 2/2) = 0
FAQ Section for NH3 Lewis Structure
Why doesn’t NH3 have a double bond?
Ammonia does not form a double bond because nitrogen, although having five valence electrons, needs only three to satisfy the octet rule, and three single bonds (to three hydrogen atoms) are enough. A double bond would leave nitrogen with only six electrons, not satisfying the octet rule optimally.
What is the importance of the lone pair in NH3?
The lone pair on nitrogen is crucial because it affects the shape and polarity of the NH3 molecule. It creates a trigonal pyramidal shape and makes the molecule polar, as the lone pair causes an electron-density imbalance around the nitrogen.
How can I remember NH3’s Lewis structure?
A simple mnemonic could be “Nitrogen has a triple desire for bonds but only needs three.” Since nitrogen can form three single bonds with hydrogen atoms and still have a lone pair, it makes the structure easier to remember. Just visualize nitrogen holding three single bonds and one lone pair.
We hope this detailed guide helps you understand and master the NH3 Lewis structure with practical tips and step-by-step instructions to make your learning journey smooth and effective!