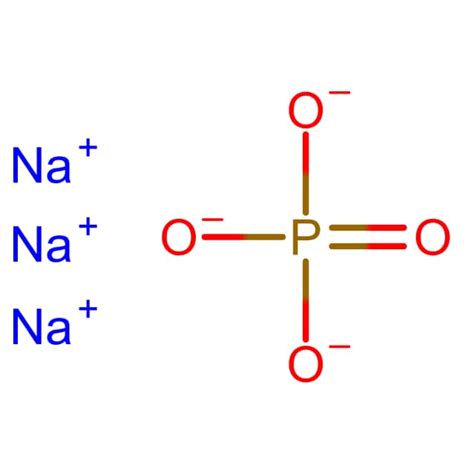
Phosphate is a critical component in many industrial and biological processes, and one of its common forms is Sodium Phosphate (Na3PO4). Understanding the molar mass of Na3PO4 is essential for anyone working in chemistry, biochemistry, or environmental science. This article provides a focused and comprehensive guide to understanding the molar mass of Na3PO4, complete with expert insights, evidence-based statements, and practical examples.
To begin with, Na3PO4, also known as trisodium phosphate, is a widely used chemical in detergents, cleaners, and food processing. Its precise molar mass is crucial for these applications, ensuring accurate formulation and effectiveness. By knowing the molar mass, professionals can precisely measure and mix the chemical in various applications, making it an indispensable piece of knowledge.
Key insights box:
Key Insights
- Primary insight with practical relevance: Calculating the molar mass of Na3PO4 ensures precise formulation in industrial and laboratory settings.
- Technical consideration with clear application: Accurate molar mass computation helps in determining the correct amount of sodium phosphate needed for specific chemical reactions.
- Actionable recommendation: Always cross-check your calculated molar mass of Na3PO4 with reliable chemical databases to confirm accuracy.
The molar mass of a compound is the sum of the atomic masses of all the atoms in its molecular formula. To calculate the molar mass of Na3PO4, one must consider the atomic masses of sodium (Na), phosphorus (P), and oxygen (O). Sodium has an atomic mass of approximately 22.99 g/mol, phosphorus has an atomic mass of about 30.97 g/mol, and oxygen has an atomic mass of roughly 16.00 g/mol.
Here’s a breakdown for Na3PO4:
- For sodium (Na): Na appears three times in the formula, so 3 * 22.99 g/mol = 68.97 g/mol
- For phosphorus (P): P appears once in the formula, so 1 * 30.97 g/mol = 30.97 g/mol
- For oxygen (O): O appears four times in the formula, so 4 * 16.00 g/mol = 64.00 g/mol
Adding these together, the molar mass of Na3PO4 is 68.97 g/mol + 30.97 g/mol + 64.00 g/mol = 163.94 g/mol.
This detailed calculation highlights the importance of accuracy in chemical formulations. For instance, in biochemical labs, precise molar mass knowledge ensures accurate dilutions and concentrations when preparing solutions of sodium phosphate for enzyme reactions or cell culture media.
The relevance of molar mass extends to industrial applications as well. In cleaning products, knowing the exact amount of Na3PO4 is crucial for formulation efficacy. An incorrect calculation could lead to ineffective cleaning agents or, worse, environmental hazards due to chemical imbalance.
In biochemistry, the molar mass of Na3PO4 plays a vital role in determining reaction stoichiometry. Accurate molar mass allows for precise calculation of reactants and products in biochemical pathways, essential for research and development.
FAQ section:
Is Na3PO4 dangerous to handle?
While Na3PO4 is generally safe when handled correctly, it should be treated with care. It is mildly irritating to the skin and eyes. Always use personal protective equipment (PPE) and ensure proper ventilation when working with it.
How do I store Na3PO4?
Store Na3PO4 in a cool, dry place, away from incompatible substances like acids. Keep the container tightly sealed to prevent moisture absorption and degradation.
Understanding the molar mass of Na3PO4 is crucial for both academic and industrial success. With a precise value of 163.94 g/mol, professionals can ensure accurate formulations in various applications, from biochemistry to industrial cleaning. Always remember to verify calculations and adhere to safety guidelines when handling this important chemical.