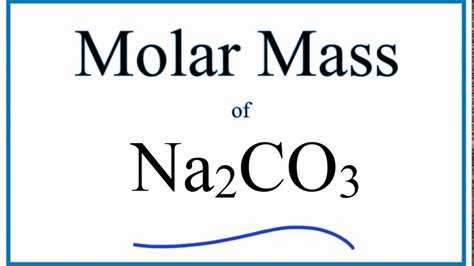Understanding the molar mass of sodium carbonate (Na2CO3) is essential for various scientific applications, from laboratory experiments to industrial processes. The precise calculation of this compound’s molar mass not only aids in stoichiometric calculations but also helps in comprehending its chemical behavior in different reactions. This article delves into the significance of Na2CO3 molar mass, backed by practical insights and real examples.
Key Insights
- Primary insight with practical relevance: Accurate molar mass calculation allows for precise measurement in chemical reactions.
- Technical consideration with clear application: Knowing the molar mass of Na2CO3 enables accurate formulation of chemical solutions.
- Actionable recommendation: Use the calculated molar mass in lab settings for exact measurements.
Why Molar Mass Matters
Molar mass is a fundamental concept in chemistry, representing the mass of one mole of a substance. For sodium carbonate, calculating the molar mass involves summing the atomic masses of its constituent elements: sodium (Na), carbon ©, and oxygen (O). Each atom’s contribution to the compound’s weight is crucial in determining how much material you need for specific reactions or solutions. Understanding Na2CO3 molar mass ensures that chemists and researchers can make accurate measurements, leading to more reliable experimental outcomes.
Breaking Down the Calculation
To calculate the molar mass of Na2CO3, we need to consider the atomic masses of each element in the formula. Here’s a step-by-step guide:
Step 1: Identify the atomic masses of each element:
- Sodium (Na): Approximately 22.99 g/mol - Carbon ©: Approximately 12.01 g/mol - Oxygen (O): Approximately 16.00 g/molStep 2: Multiply the atomic mass of each element by its occurrence in the formula:
- Sodium: 2 atoms × 22.99 g/mol = 45.98 g/mol - Carbon: 1 atom × 12.01 g/mol = 12.01 g/mol - Oxygen: 3 atoms × 16.00 g/mol = 48.00 g/molStep 3: Sum these values to obtain the total molar mass:
- Total molar mass of Na2CO3 = 45.98 g/mol (Na) + 12.01 g/mol © + 48.00 g/mol (O) = 105.99 g/molThe precision in this calculation ensures that the correct amount of sodium carbonate is used in reactions, leading to the desired results without excess or deficit.
Application in Laboratory Settings
In laboratory settings, the molar mass of Na2CO3 is instrumental in several scenarios. For example, when preparing a sodium carbonate solution for titrations or other analytical procedures, knowing the exact molar mass allows for precise dilution calculations. Miscalculating the molar mass could result in inaccurate concentrations, compromising the experiment’s validity.
Consider an experiment requiring 0.1 moles of sodium carbonate dissolved in water to make 1 liter of solution. With a molar mass of 105.99 g/mol, we calculate the necessary mass as follows: 0.1 moles × 105.99 g/mol = 10.599 grams of Na2CO3.
Accurate preparation like this underscores the importance of understanding the compound’s molar mass.
What is the significance of knowing Na2CO3 molar mass?
Knowing the molar mass of Na2CO3 is crucial for accurate stoichiometric calculations, precise measurement in chemical reactions, and the formulation of accurate chemical solutions.
Can inaccuracies in Na2CO3 molar mass affect experimental results?
Yes, inaccuracies in molar mass can lead to incorrect concentrations, affecting the reliability and outcomes of chemical experiments and analyses.
In conclusion, the molar mass of sodium carbonate is a critical piece of information for any chemist or researcher. The detailed calculation ensures accurate and precise applications in both theoretical and practical chemical endeavors, reinforcing the importance of exactness in scientific measurements.