Understanding the intricacies of biological molecules is essential for many fields, from biochemistry to medicine, and it’s especially crucial for anyone delving into the structure and function of living organisms. Today, we’re going to demystify one of the most versatile and vital classes of biomolecules: lipids. We’ll focus on the building blocks of lipids, known as monomers, and how they come together to perform myriad functions in our bodies and other living organisms.
Whether you're a student, a researcher, or just someone curious about the inner workings of biology, this guide will provide you with practical, actionable advice to understand and apply knowledge about lipid monomers. By the end of this guide, you’ll have a solid foundation in lipid biology and the ability to recognize the critical roles these molecules play.
Understanding Lipid Monomers: A Problem-Solution Approach
Many people struggle to grasp the complexity of lipids due to their seemingly vast diversity and the broad range of functions they perform. Lipids are a varied group of organic compounds that are insoluble in water but soluble in organic solvents. They include fats, oils, waxes, certain vitamins, hormones, and most of the non-protein membranes of cells, as well as a major component of all cell membranes. They are crucial for energy storage, structural integrity, and signaling in the body. However, where to start and how to piece together their intricate functions can be daunting. This guide aims to simplify that process by breaking down the fundamental monomers that form these essential molecules, thus making their study approachable and engaging.
Quick Reference: Getting Started with Lipid Monomers
Quick Reference
- Immediate action item with clear benefit: Identify common fatty acids in your diet and understand their roles in health.
- Essential tip with step-by-step guidance: Learn how to recognize and classify different types of lipids by their structural features.
- Common mistake to avoid with solution: Don’t confuse lipids with proteins or carbohydrates. Understand that while they share some cellular roles, their structures and functions are distinct.
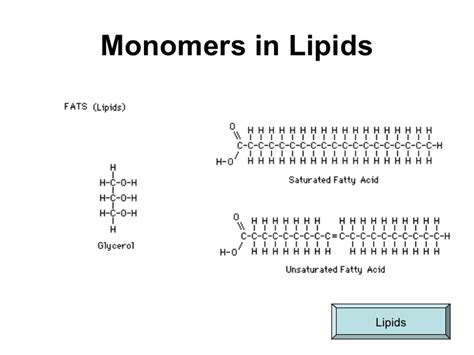
Breaking Down Fatty Acids: The Basic Monomers
Fatty acids are the simplest form of lipid monomers. These are long hydrocarbon chains terminated by carboxyl groups. Fatty acids can vary in length and the degree of saturation of their hydrocarbon chains. Understanding these differences is crucial for grasping how they will interact to form more complex lipids.
Length and Structure: Fatty acids can range from very short (two to three carbons) to very long (more than twenty carbons). Short-chain fatty acids (like acetic acid) are common in microbial metabolism, while longer-chain fatty acids (like stearic acid) are common in animal fats. The most common fatty acids found in human diet and body tissues are palmitic acid (16 carbons) and oleic acid (18 carbons). Saturation Levels: The degree of saturation refers to how many double bonds are present in the fatty acid. Saturated fatty acids have no double bonds between the carbon atoms, while unsaturated fatty acids contain one or more double bonds. The presence of these double bonds introduces kinks in the carbon chain, affecting how fatty acids pack together. For instance, unsaturated fats remain liquid at room temperature due to these kinks, while saturated fats tend to be solid.
Practical Steps to Understanding Fatty Acids
Step 1: Observe your diet. Identify and note the types of fats you consume. Are they saturated or unsaturated? How do these fats affect your health?
Step 2: Experiment with classifying fats. Try substituting saturated fats with unsaturated ones and observe any changes in texture or health benefits.
Step 3: Research and document the impact of different fatty acids on human health. Understanding which types are beneficial and which can be harmful is essential for maintaining a healthy diet.
Step 4: Engage in educational activities. Attend workshops or online courses focused on lipid biochemistry to deepen your understanding.
Constructing Complex Lipids from Fatty Acids
While fatty acids are fundamental, their real power shines when they combine to form more complex lipids like triglycerides, phospholipids, and glycolipids. These are the building blocks for cell membranes, energy storage forms, and signaling molecules.
Triglycerides: Storing Energy Efficiently
Triglycerides are formed when three fatty acids are esterified to a glycerol backbone. The glycerol molecule provides a central core, and each of its three hydroxyl groups links to a fatty acid chain, creating a triglyceride molecule.
Step-by-Step Construction:
- Identify glycerol, a simple sugar alcohol, which will serve as the core structure.
- Choose three fatty acids; their length and saturation level will influence the properties of the resulting triglyceride.
- Form ester bonds between the hydroxyl groups of glycerol and the carboxyl groups of the fatty acids.
- The result is a triglyceride molecule ready to be stored in adipose tissue or used as an energy source.
Phospholipids: The Architects of Membranes
Phospholipids are crucial for the structure of cell membranes. They consist of a glycerol backbone, two fatty acid tails, and a phosphate group attached to a nitrogen-containing group.
Step-by-Step Construction:
- Begin with a glycerol backbone.
- Attach two fatty acids through ester linkages to the first two hydroxyl groups of glycerol.
- Add a phosphate group linked to a nitrogenous group through an ester bond to the third hydroxyl group.
- The resulting phospholipid has hydrophilic (water-attracting) and hydrophobic (water-repelling) regions, making it perfect for forming bilayers in cell membranes.
Practical FAQ: Demystifying Lipid Monomers
Why are lipids considered hydrophobic?
Lipids are primarily composed of hydrocarbon chains, which are non-polar and thus repel water (hydrophobic). This property allows them to form barriers, like cell membranes, and to serve as energy stores, since they can pack tightly without needing to hydrate. The hydrophobic nature is fundamental to their biological roles, from creating lipid bilayers that form the basis of cell membranes to storing energy efficiently in adipose tissue.
What is the difference between saturated and unsaturated fats?
Saturated fats contain no double bonds between the carbon atoms in their hydrocarbon chains, leading to a straight structure that allows them to pack closely together, often resulting in a solid state at room temperature. Examples include butter and lard. Unsaturated fats contain one or more double bonds, which introduce kinks in their structure, preventing close packing and keeping them liquid at room temperature. These are typically found in plant oils like olive oil and fish. The health impacts of saturated fats often include higher risks for heart disease when consumed in excess, while unsaturated fats are generally considered healthier and can lower bad cholesterol levels.
Final Tips and Best Practices
Here are some final tips to remember as you delve deeper into the world of lipid monomers:
- Stay curious and question everything. The more you understand about lipid structures, the better you can apply this knowledge to practical situations.
- Use online resources and textbooks to keep learning. Knowledge in this field is always expanding, so stay updated.
- Always relate your studies to real-world examples. For instance, think about how dietary choices impact overall health through the lens of lipid biochemistry.
- Engage with the scientific community through forums or professional networks to share ideas and discoveries.
This guide has provided you with a comprehensive understanding of lipid monomers, their roles, and how they form complex lipids. Armed with this knowledge, you’re better prepared to tackle advanced topics in lipid biochemistry and appreciate the nuanced roles these molecules play in biology and medicine.