Understanding the molecular weight of carbon is vital for numerous scientific applications, ranging from material science to biochemistry. This foundational concept underpins many areas of research, yet it’s often misunderstood or overlooked in broader chemical contexts. By delving into carbon’s molecular weight, we unlock insights that are not only academically interesting but also practically indispensable.
Key insights box:
Key Insights
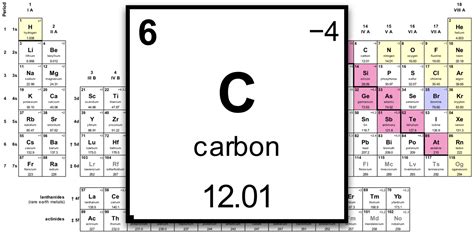
- The standard atomic weight of carbon © is 12.011 amu, pivotal in calculating molecular weights.
- Understanding carbon’s isotopic distribution aids in accurate molecular composition analysis.
- Practical application includes identifying contaminants in biochemical assays.
Carbon’s ubiquitous presence in organic chemistry and biochemistry necessitates a robust comprehension of its molecular weight. Carbon atoms form the backbone of organic compounds, making their molecular weight essential for defining the structure and behavior of complex molecules. The commonly recognized atomic weight of carbon is approximately 12.011 atomic mass units (amu). This figure arises from the weighted average of all carbon isotopes found on Earth, primarily carbon-12 and carbon-13, along with a trace amount of carbon-14.
Carbon-12 serves as the international standard for atomic mass scale, meaning that one atom of carbon-12 is defined as exactly 12 amu. This standardization is crucial when calculating the molecular weights of organic compounds, where carbon atoms typically play a central role. For example, glucose (C6H12O6) has a molecular weight calculated by summing the weights of six carbon atoms (6 x 12.011 amu), twelve hydrogen atoms (12 x 1.008 amu), and six oxygen atoms (6 x 16.00 amu), equating to 180.16 amu. This precise measurement aids in understanding metabolic pathways and energy production in living organisms.
An often overlooked aspect is the isotopic distribution of carbon. Natural carbon is approximately 98.9% carbon-12, 1.1% carbon-13, and traces of carbon-14. This distribution is not just academic—it’s crucial for accurate molecular composition analysis. Carbon-14, in particular, has significant applications in radiocarbon dating, a technique used to determine the age of organic materials. Knowing the relative abundance of carbon isotopes enhances the precision of these analyses and is vital in fields like archaeology and geology.
This nuanced understanding of carbon’s isotopic composition and molecular weight extends beyond academic curiosity. In practice, it has implications for identifying contaminants in biochemical assays. Accurate molecular weight calculations ensure that researchers can distinguish between naturally occurring compounds and impurities, thus maintaining the integrity of experimental results. For instance, in drug development, precise molecular weights help in identifying unintended by-products or contaminants that could interfere with the efficacy and safety of pharmaceutical products.
FAQ section:
Why is carbon’s atomic weight not exactly 12?
Carbon’s atomic weight is an average calculated from its naturally occurring isotopes: carbon-12, carbon-13, and carbon-14. The atomic weight of 12.011 amu accounts for their relative abundances.
How do isotopes of carbon impact molecular weight calculations?
Isotopes affect molecular weight by contributing different atomic masses. For example, carbon-13 has an atomic weight of about 13.003 amu, slightly more than carbon-12, which alters the molecular weight calculations of compounds that include carbon-13.
In summary, the molecular weight of carbon is not just a numerical value but a critical factor influencing the accuracy of molecular composition analyses and practical applications across various scientific fields. Accurate knowledge of carbon’s atomic weight and isotopic distribution supports high-quality research and reliable technological advancements.