Understanding the Molecular Shape of CO2: Unveil Secrets of Carbon Dioxide Geometry
Understanding the molecular shape of CO2 is pivotal for grasping various chemical and physical properties of carbon dioxide. This molecule’s geometry underpins its unique behavior in the atmosphere and its implications for global climate change. Carbon dioxide (CO2), comprised of one carbon atom bonded to two oxygen atoms, provides a fascinating insight into molecular structure, bonding, and the significance of geometry in chemical behavior.
Why Molecular Geometry Matters
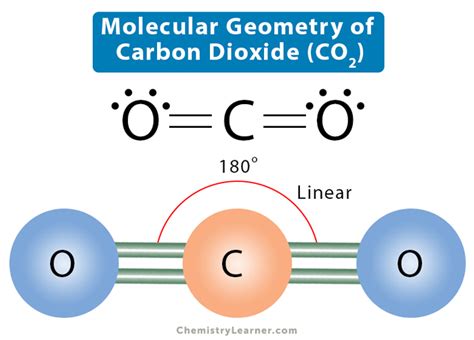
The molecular shape of CO2 is determined by its linear geometry, a fundamental aspect of its structure. The carbon atom forms double bonds with each of the two oxygen atoms. This linear arrangement is dictated by the electron pair repulsion theory, where valence shell electrons repel each other to achieve maximum separation, resulting in a linear shape with a bond angle of 180 degrees. This geometry is crucial because it determines how CO2 interacts with other molecules, its physical properties, and its role in atmospheric processes.Key Insights
- Primary insight with practical relevance: CO2's linear molecular geometry ensures efficient energy absorption in the infrared spectrum, contributing to greenhouse effects.
- Technical consideration with clear application: The linear structure influences CO2's inability to form hydrogen bonds, which impacts its solubility and boiling point compared to other similar molecules.
- Actionable recommendation: To better understand CO2's environmental impact, focus on its molecular geometry to comprehend its role in atmospheric retention of heat.
Valence Shell Electron Pair Repulsion (VSEPR) Theory
The VSEPR theory elucidates the molecular shape of CO2. According to this theory, the carbon atom has four valence electrons. Two of these electrons are shared with each oxygen atom forming double bonds. There are no lone pairs of electrons on the carbon atom in a CO2 molecule, resulting in a linear molecular shape. This is the simplest way to arrange these electrons to minimize repulsion, resulting in a molecule where the oxygen atoms are directly across from each other.Impact on Chemical Behavior and Environmental Implications
CO2’s linear structure is fundamental to its chemical reactivity and environmental impact. As a non-polar molecule due to its symmetrical shape, CO2 does not engage in hydrogen bonding, which affects its physical properties such as lower boiling and melting points. This non-polarity means it does not mix well with water, which is polar, explaining its limited solubility in aqueous environments.Moreover, CO2’s linear molecular geometry allows it to absorb infrared radiation efficiently, trapping heat in the Earth’s atmosphere and contributing to the greenhouse effect. This absorption is significant because it involves the vibrations of the double bonds between carbon and oxygen atoms, a phenomenon central to its role in global warming.
What is the bond angle in CO2?
The bond angle in CO2 is 180 degrees due to its linear geometry.
How does CO2’s molecular shape affect its environmental impact?
CO2’s linear and non-polar molecular shape allows it to efficiently absorb infrared radiation, contributing to the greenhouse effect and global warming.
In summation, understanding the molecular shape of CO2 through the lens of its linear geometry provides critical insights into its chemical properties and environmental impacts. This knowledge underscores the importance of molecular geometry in chemistry and its broader implications for climate science.