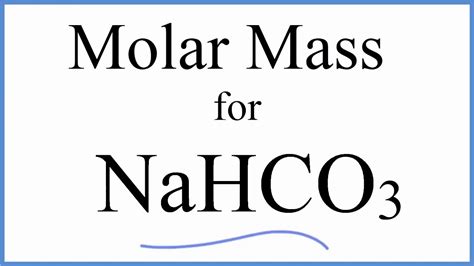Understanding the molecular mass of sodium bicarbonate, also known as baking soda, is essential for anyone working in chemistry, cooking, or even DIY projects that require precise measurements. The molecular mass (or molar mass) of sodium bicarbonate, represented as NaHCO3, might seem complex at first glance, but with a step-by-step approach, it becomes straightforward and easy to grasp. This guide will walk you through the problem-solving process of calculating molecular mass, providing actionable advice and practical examples along the way.
The Importance of Molecular Mass
The molecular mass of a compound like NaHCO3 is crucial for various applications. In chemistry, it’s fundamental for understanding stoichiometry in reactions, calculating the amount of substance involved, and ensuring accurate formulation in pharmaceuticals or food products. In cooking, especially baking, precise measurements are key to achieving desired outcomes in recipes. For DIY enthusiasts, knowing the molecular mass can aid in creating perfect mixtures for cleaning or art projects. Essentially, understanding and calculating molecular mass helps solve real-world problems efficiently and accurately.
Quick Reference
Quick Reference
- Immediate action item: Calculate the atomic masses of each element in NaHCO3 and sum them up.
- Essential tip: Use the periodic table to find the atomic mass of each element: Sodium (Na), Hydrogen (H), Carbon ©, and Oxygen (O).
- Common mistake to avoid: Forgetting to include all elements in the compound, especially oxygen, which often adds significant mass.
How to Calculate the Molecular Mass of NaHCO3
Calculating the molecular mass involves adding the atomic masses of all atoms in the compound. Here’s a detailed guide to ensure you get it right.
Step-by-Step Calculation
Follow these steps to calculate the molecular mass of NaHCO3 accurately:
- Identify the elements: NaHCO3 contains Sodium (Na), Hydrogen (H), Carbon (C), and Oxygen (O).
- Find atomic masses: Use the periodic table to find the atomic mass for each element. For NaHCO3:
- Sodium (Na): 22.99 g/mol
- Hydrogen (H): 1.01 g/mol
- Carbon (C): 12.01 g/mol
- Oxygen (O): 16.00 g/mol (and there are three oxygen atoms)
- Calculate the mass contribution of each element:
- Sodium (Na): 1 atom × 22.99 g/mol = 22.99 g/mol
- Hydrogen (H): 1 atom × 1.01 g/mol = 1.01 g/mol
- Carbon (C): 1 atom × 12.01 g/mol = 12.01 g/mol
- Oxygen (O): 3 atoms × 16.00 g/mol = 48.00 g/mol
- Sum the masses: Add up the individual contributions to get the total molecular mass of NaHCO3.
Detailed How-To Sections
Breaking Down Each Element’s Contribution
To ensure clarity, let’s break down each element’s contribution to the molecular mass of NaHCO3:
- Sodium (Na): As the least massive element in NaHCO3, sodium still plays a significant role in its molecular mass. The atomic mass of sodium is 22.99 g/mol. Since there’s only one sodium atom in each molecule of NaHCO3, its contribution to the molecular mass is straightforward: 22.99 g/mol.
- Hydrogen (H): Hydrogen has an atomic mass of 1.01 g/mol. With only one hydrogen atom per molecule of NaHCO3, its contribution is minimal but essential. Simply multiply 1.01 g/mol by 1 to get 1.01 g/mol.
- Carbon ©: Carbon’s atomic mass is 12.01 g/mol. Since there is only one carbon atom in NaHCO3, its contribution is direct: 12.01 g/mol. This element adds a modest but significant mass to the compound.
- Oxygen (O): Oxygen has an atomic mass of 16.00 g/mol, but because there are three oxygen atoms in NaHCO3, its contribution is more substantial. Multiply the atomic mass by the number of oxygen atoms: 16.00 g/mol × 3 = 48.00 g/mol.
Putting It All Together
To calculate the total molecular mass of NaHCO3, add up the contributions from each element:
- Sodium: 22.99 g/mol
- Hydrogen: 1.01 g/mol
- Carbon: 12.01 g/mol
- Oxygen: 48.00 g/mol
Summing these values gives:
22.99 g/mol + 1.01 g/mol + 12.01 g/mol + 48.00 g/mol = 84.01 g/mol
Thus, the molecular mass of NaHCO3 is 84.01 g/mol.
Practical Examples and Applications
Let’s explore some practical scenarios where knowing the molecular mass of NaHCO3 is crucial:
Chemistry Laboratory
In a lab setting, precise measurements are critical. When preparing a solution of sodium bicarbonate for a titration experiment, knowing its molecular mass ensures accurate concentration calculations. For instance, if you need 0.1 moles of NaHCO3 for a reaction, you’ll use the molecular mass to determine the weight needed: 0.1 moles × 84.01 g/mol = 8.401 grams.
Baking Recipes
Baking requires precision to achieve the perfect texture and rise. For instance, if a recipe calls for 1 teaspoon of baking soda, understanding its molecular mass can help in scaling recipes accurately if using weight instead of volume. Given that 1 teaspoon of baking soda is approximately 4.44 grams, you can determine the equivalent in moles: 4.44 grams / 84.01 g/mol ≈ 0.0528 moles.
DIY Cleaning Solutions
Many DIY cleaning solutions involve sodium bicarbonate. Suppose you’re creating a cleaning paste to polish silver. You’ll need to mix NaHCO3 with other ingredients. Knowing the molecular mass allows you to balance the mixture precisely, ensuring effectiveness without excess or shortage.
Practical FAQ
What is the importance of knowing the molecular mass of NaHCO3 in everyday applications?
Knowing the molecular mass of NaHCO3 is essential in various everyday applications, from chemistry labs to home baking and DIY projects. It allows for precise measurements and accurate formulation of solutions or recipes. Whether you’re conducting a lab experiment, baking with exact ratios, or creating