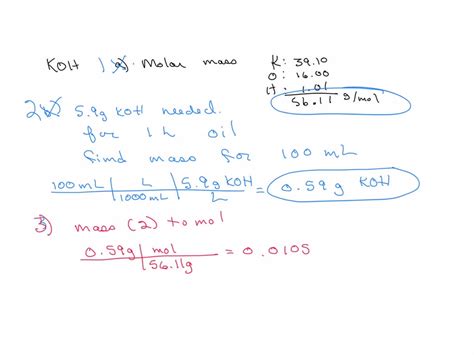Understanding the molar weight of potassium hydroxide (KOH) is essential for chemists and chemical engineers who deal with this compound in various applications. Knowing its molar weight can help with precise formulation in chemical reactions, accurate dosage in chemical processes, and effective management of chemical inventory. Here, we provide a comprehensive guide that goes beyond basic facts to offer actionable advice, practical solutions, and expert insights.
Why Molar Weight Matters for KOH
The molar weight of a substance is a fundamental parameter that determines the amount needed for specific chemical reactions. For potassium hydroxide (KOH), it plays a pivotal role in various industrial and laboratory settings. Miscalculating the molar weight can lead to errors in chemical formulation, inefficient use of resources, and safety concerns. This guide focuses on equipping you with the knowledge and tools to determine and use the molar weight of KOH with confidence.
Quick Reference Guide for Molar Weight of KOH
Quick Reference
- Immediate action item with clear benefit: Use a calibrated balance to weigh your KOH samples for precise molar calculations.
- Essential tip with step-by-step guidance: To find the molar weight of KOH, sum the atomic weights of potassium (K), oxygen (O), and hydrogen (H). Potassium’s atomic weight is approximately 39.10 g/mol, oxygen is 16.00 g/mol, and hydrogen is 1.01 g/mol. The total is 56.11 g/mol.
- Common mistake to avoid with solution: A common pitfall is ignoring the molecular formula. Always ensure you’re calculating for KOH, not KH₂O. Mistaking the formula can lead to incorrect molar weight calculations.
Detailed Calculation of Molar Weight of KOH
Let’s dive deeper into how to calculate the molar weight of KOH step-by-step. Understanding these steps can help you verify the weight and ensure accuracy in various chemical applications.
Step-by-Step Guide to Calculating Molar Weight of KOH
To determine the molar weight of KOH, follow these steps:
- Identify the molecular composition: Potassium hydroxide, commonly known as KOH, consists of potassium (K), oxygen (O), and hydrogen (H) atoms.
- Find atomic weights: Look up the atomic weights of each element:
- Potassium (K): 39.10 g/mol
- Oxygen (O): 16.00 g/mol
- Hydrogen (H): 1.01 g/mol
- Calculate the total molar weight: Sum the atomic weights of all atoms in the formula.
Molar weight of KOH = 39.10 g/mol (K) + 16.00 g/mol (O) + 1.01 g/mol (H) = 56.11 g/mol
By knowing these steps, you can ensure that you’re calculating the molar weight of KOH correctly and applying this knowledge to your specific chemical tasks.
Verification Through Formulae and Standards
For verification, you can cross-check using standard molar weight tables available in chemistry textbooks and online databases. These are generally reliable and can help confirm your calculations.
Practical Considerations
When working in a lab or industrial setting, it’s vital to take into account practical considerations:
- Always use high-precision instruments for weighing KOH samples.
- Keep the environment dry to avoid KOH’s hygroscopic nature, which makes it absorb moisture from the air.
- Store KOH in tightly sealed containers to maintain its purity and prevent degradation.
Practical FAQ: Molar Weight of KOH
How is the molar weight of KOH relevant in chemical reactions?
The molar weight of KOH is crucial in chemical reactions for several reasons:
- Stoichiometric calculations: It helps in determining the correct stoichiometric amounts of reactants and products in chemical equations.
- Concentration preparation: For preparing solutions of known concentration, knowing the molar weight allows for accurate dissolution calculations.
- Reaction efficiency: Ensures precise measurements, which can prevent overuse or underuse of reactants, thus optimizing reaction efficiency and minimizing waste.
What happens if the molar weight of KOH is not used correctly?
Misuse of the molar weight of KOH can lead to several problems:
- Reaction imbalances: Can cause the reaction to go off-balance, leading to incomplete reactions or the formation of unwanted by-products.
- Inaccurate results: Can lead to incorrect experimental results, which undermines the reliability of the study or industrial process.
- Economical losses: Miscalculations can result in inefficient use of resources and increased costs due to excess reactants or waste.
Advanced Tips for Handling KOH in Industrial Applications
When working with KOH in an industrial setting, the following advanced tips can enhance your handling and usage:
- Batch consistency: Ensure uniformity in the batches of KOH to avoid variations in molar weight due to impurities or deviations in production.
- Quality control: Implement stringent quality control measures to maintain the purity of KOH and avoid contamination that might alter its weight or properties.
- Safety protocols: Always follow safety guidelines for handling KOH. It is caustic and can cause severe burns upon contact with skin or damage to surfaces. Proper PPE (personal protective equipment) and safety training are mandatory.
By adhering to these advanced practices, you can maintain high standards in the handling and application of potassium hydroxide in industrial settings.
Practical Applications of KOH
Potassium hydroxide has a wide range of applications across various industries:
- Soap production: It is a key ingredient in the saponification process for making soap.
- De-icing: Used in de-icing solutions due to its ability to lower the freezing point of water.
- Batteries: Utilized in alkaline batteries as an electrolyte.
- Catalyst: Acts as a catalyst in many organic reactions, including the conversion of vegetable oils into biodiesel.
Understanding its molar weight aids in accurately dosing it in these applications, ensuring effective performance and safety.
Case Study: KOH in Battery Production
Let’s look at a practical example of KOH’s application in alkaline battery production:
In battery manufacturing, the precise amount of KOH is essential for the optimal functioning of the battery. Suppose the manufacturing process calls for a specific concentration of KOH in the electrolyte solution. Accurate calculation and measurement based on its molar weight ensure the right ratio, leading to efficient battery performance and longevity.
Conclusion
The molar weight of KOH, though a basic concept, plays a critical role in various chemical and industrial processes. By mastering the calculation, verification, and practical application of KOH’s molar weight, you enhance your efficiency and safety in handling this versatile compound. This guide aims to provide you with the knowledge and tools to approach your tasks with confidence and precision.
This guide combines detailed instructions, practical examples, and advanced tips to ensure that chemists and chemical engineers can confidently use KOH in their applications. By integrating this knowledge into your workflow, you’ll achieve accurate formulations, better process management, and overall improved outcomes in your work.