Understanding the molar mass of elements like sulfur (S) is a crucial aspect of chemistry, and it forms the foundation for many advanced concepts in the field. This guide will help you navigate through the process of calculating the molar mass of sulfur in a step-by-step manner. Whether you are a student, a chemist, or simply someone curious about the basics of chemistry, this guide aims to provide you with clear, actionable advice and practical solutions to understand and apply the concept effectively. By the end of this guide, you will not only know how to calculate the molar mass of sulfur but also understand why it's an essential concept in chemistry.
Understanding the Need for Calculating Molar Mass
The molar mass of an element is defined as the mass of one mole of that element. Knowing the molar mass is essential for various chemical calculations such as determining the amount of a substance needed for a reaction, calculating reaction yields, and understanding stoichiometry. In chemistry, precise measurements are vital, and molar mass provides the bridge between the atomic world and the macroscopic quantities we use in laboratories and everyday life. The molar mass of sulfur, therefore, is not just a number but a gateway to understanding chemical reactions at a deeper level.
Why Is It Important?
Calculating the molar mass is fundamental for several reasons:
- Reaction Stoichiometry: It helps in balancing chemical equations and ensuring the correct proportions of reactants and products.
- Precise Measurements: Knowing the molar mass allows for accurate measurements of substances in experiments.
- Understanding Molecular Composition: It provides insight into the composition and structure of molecules.
Quick Reference
Quick Reference
- Immediate action item: Look up the atomic mass of sulfur on the periodic table.
- Essential tip: Remember, the atomic mass unit (amu) is a tiny fraction of a gram per mole; 1 amu ≈ 1.66 x 10^-24 grams.
- Common mistake to avoid: Confusing atomic mass with atomic weight, which includes isotopic variations.
Calculating the Molar Mass of Sulfur
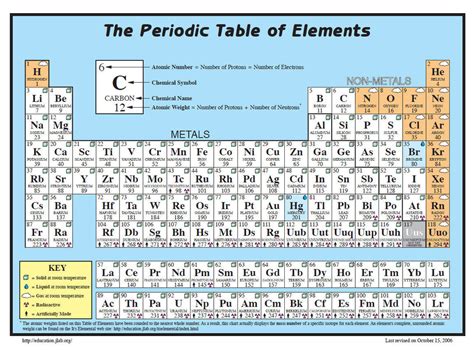
To calculate the molar mass of sulfur, we start by understanding the periodic table. The atomic mass of sulfur (S) is given directly on the periodic table, usually accompanied by a value that accounts for the natural abundance of its isotopes. The atomic mass of sulfur is approximately 32.06 amu (atomic mass units). To convert this into grams per mole (g/mol), we need to understand that 1 amu is equivalent to 1.66 x 10^-24 grams, but for practical purposes in chemistry, we use the periodic table’s atomic mass directly as it already represents grams per mole for one mole of atoms.
For sulfur, the molar mass is approximately 32.06 grams per mole. This means that one mole of sulfur atoms weighs 32.06 grams. This calculation is straightforward because the atomic mass unit directly correlates to grams per mole for the purposes of stoichiometry and chemical calculations. Let's delve deeper into the steps to ensure a complete understanding.
Step-by-Step Calculation
Here’s how to calculate the molar mass of sulfur in a detailed manner:
- Identify the Atomic Mass: From the periodic table, note the atomic mass of sulfur. It's approximately 32.06 amu. This value is often rounded to 32.06 g/mol for simplicity in chemical calculations.
- Conversion of Atomic Mass Units to Grams per Mole: Although the conversion factor (1 amu ≈ 1.66 x 10^-24 grams) is used in atomic calculations, for molar mass, we consider the atomic mass directly in grams per mole because it has been averaged over naturally occurring isotopes. This step simplifies our calculations.
- Calculation: Since the atomic mass for sulfur (32.06 amu) already represents the mass in grams per mole for one mole of sulfur atoms, no additional conversion is needed. Therefore, the molar mass of sulfur is exactly 32.06 grams per mole.
Applying the Concept in Real-World Scenarios
Knowing the molar mass of sulfur can help in various practical applications. Let’s explore a few scenarios where this knowledge is directly applicable:
Preparing Chemical Solutions
When preparing a chemical solution in a lab, knowing the molar mass of a solute like sulfur in a solvent allows for accurate calculations of the amount of solute needed to achieve a desired concentration. For instance, if you need to prepare a 0.1 M solution of sulfur in water, knowing that the molar mass is 32.06 grams per mole means you need 32.06 grams of sulfur for every liter of solution.
Analyzing Reactions
In analytical chemistry, determining the amount of sulfur in a compound is crucial. For example, if you have a compound that contains sulfur and you need to determine its mass percentage, knowing the molar mass of sulfur allows you to calculate the total mass of sulfur in the compound and compare it to the compound’s total mass.
Educational Demonstrations
Teachers and educators can use the concept of molar mass to demonstrate basic principles of chemistry in an engaging and practical way. For instance, calculating the molar mass of sulfur and comparing it to other elements helps students understand the diversity and uniformity in atomic masses, leading to a deeper comprehension of chemical principles.
Practical FAQ
What is the difference between atomic mass and molar mass?
Atomic mass refers to the mass of a single atom of an element, typically measured in atomic mass units (amu). It represents the weighted average mass of the element’s isotopes. Molar mass, on the other hand, refers to the mass of one mole of an element, which is the amount of substance containing as many entities (atoms, molecules, ions, etc.) as there are atoms in 12 grams of carbon-12. For simplicity, the atomic mass in grams per mole is numerically equal to the atomic mass in amu for elements.
Why is it important to know the molar mass of an element?
Knowing the molar mass of an element is crucial for several reasons. It allows chemists to make precise measurements of substances needed for experiments and reactions, helps in understanding the stoichiometry of chemical reactions, and is fundamental in converting between the number of atoms and grams of a substance. This knowledge is vital for everything from laboratory work to industrial applications.
Can molar mass vary for the same element?
While the molar mass for a single element generally does not vary, it can depend on the isotopic composition of the element being considered. Naturally occurring elements often consist of a mix of isotopes, each with slightly different masses. Therefore, the molar mass provided on the periodic table is an average value based on the natural abundance of these isotopes.
This guide provides a comprehensive approach to understanding the molar mass of sulfur, its calculation, and its practical applications. By mastering this concept, you enhance your ability to engage in more complex chemical analysis and experiments, thereby expanding your knowledge and capabilities in the field of chemistry.