Understanding the molar mass of Pb(NO3)2 is crucial for anyone studying chemistry, whether you’re a student tackling your first lab or a seasoned chemist needing a refresher. The molar mass of a compound is essentially the weight of a given substance in grams per mole. Calculating this for Pb(NO3)2 requires understanding each element’s atomic weight and applying a bit of simple math.
In this guide, we’ll walk you through the process of calculating the molar mass of Pb(NO3)2, with real-world examples and practical solutions to any potential confusion.
We'll provide step-by-step guidance with actionable advice, tips, and best practices to ensure you grasp this concept thoroughly. This guide aims to address common pain points and offer straightforward solutions, ensuring you can apply this knowledge confidently in any chemistry context.
Why Knowing the Molar Mass of Pb(NO3)2 Matters
Calculating the molar mass of compounds is fundamental to chemistry. Whether you’re preparing solutions for experiments or performing stoichiometric calculations, knowing the molar mass is crucial. For Pb(NO3)2, it’s especially significant because it’s a common compound in various chemical reactions and applications. Misunderstanding or miscalculating this value can lead to incorrect results, whether it’s in lab work or theoretical problem-solving.
Let’s dive into how you can determine this molar mass accurately. Understanding the basic principles behind the calculation will empower you to tackle similar problems with confidence.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Break down Pb(NO3)2 into its constituent elements: lead (Pb), nitrogen (N), and oxygen (O). This will help in calculating the molar mass step-by-step.
- Essential tip with step-by-step guidance: Use the periodic table to find the atomic weights of each element in the compound. For Pb(NO3)2, you’ll need the atomic weights of Pb, N, and O.
- Common mistake to avoid with solution: Don’t forget to multiply the atomic weights by the number of atoms of each element in the compound. In Pb(NO3)2, there’s 1 Pb, 2 N’s, and 6 O’s.
Detailed How-to Section: Calculating the Molar Mass of Pb(NO3)2
Calculating the molar mass of Pb(NO3)2 involves a straightforward but systematic approach. Here’s how to do it:
- Identify the Elements: Pb(NO3)2 is composed of lead (Pb), nitrogen (N), and oxygen (O).
- Find Atomic Weights: Obtain the atomic weights of each element from the periodic table. Here are the relevant values:
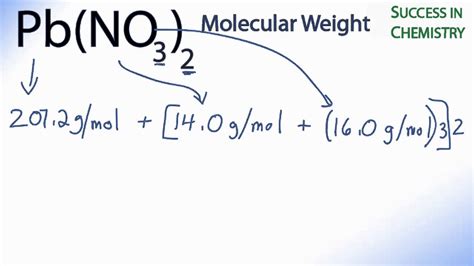
- Lead (Pb): 207.2 g/mol
- Nitrogen (N): 14.01 g/mol
- Oxygen (O): 16.00 g/mol
- Break Down the Formula: Each nitrate (NO3) group in Pb(NO3)2 contains 1 nitrogen and 3 oxygens. There are two nitrate groups in Pb(NO3)2.
- Calculate the Molar Mass: Add the atomic weights of the individual elements, accounting for the number of each type of atom in the formula.
Here’s the breakdown:
| Element | Atomic Weight | Number of Atoms | Total Weight |
|---|---|---|---|
| Pb | 207.2 g/mol | 1 | 207.2 g/mol |
| N | 14.01 g/mol | 2 | 28.02 g/mol |
| O | 16.00 g/mol | 6 | 96.00 g/mol |
To find the total molar mass of Pb(NO3)2, simply add the total weights of each element:
207.2 g/mol (Pb) + 28.02 g/mol (2N) + 96.00 g/mol (6O) = 331.22 g/mol
Thus, the molar mass of Pb(NO3)2 is 331.22 g/mol.
Practical FAQ
Why is it important to know the exact molar mass?
Knowing the exact molar mass is critical for several reasons:
- It ensures accurate preparation of solutions.
- It’s essential for stoichiometric calculations in chemical reactions.
- It helps in determining the amount of a substance needed for a given reaction.
For Pb(NO3)2, precision in molar mass calculation guarantees that you use the correct amount of the compound in lab experiments, which directly impacts the success of the reaction.
What should I do if I make a mistake in calculating the molar mass?
If you find yourself making mistakes, here’s what you can do:
- Double-check each atomic weight you’ve used for accuracy.
- Verify the number of each type of atom in the formula.
- Recalculate each step carefully.
Miscalculating the molar mass can lead to incorrect results in your experiments. Always take a few extra minutes to review your work, especially in the context of lab work.
By following this detailed guide, you should now have a clear understanding of how to calculate the molar mass of Pb(NO3)2. With practical examples and tips integrated into this process, you can now apply this knowledge with confidence in any chemistry-related task.
Remember, the key to mastering this concept is practice. Make it a point to work through similar problems, and soon, calculating molar masses will become second nature.
This guide serves as a comprehensive resource for anyone needing to delve into the specifics of molar mass calculation. It not only addresses the problem-solving aspect but also provides practical examples to ensure you can apply this knowledge effectively.
Thank you for reading this guide. If you have any further questions or need additional clarification on any part, feel free to reach out or consult additional chemistry resources.