Understanding the molar mass of methanol is fundamental for anyone involved in chemical calculations, research, or industrial applications. Methanol, commonly known as wood alcohol, serves as a crucial compound in various sectors, including pharmaceuticals, fuel production, and as a chemical solvent. To fully grasp the importance of methanol’s molar mass, one must appreciate its composition and the methodical approach to calculating it.
Key Insights
- Methanol’s molar mass is essential for precise chemical calculations, especially in stoichiometry.
- The atomic weights of carbon, hydrogen, and oxygen dictate methanol’s molar mass.
- Using the correct molar mass ensures accurate conversions in chemical reactions.
Understanding Methanol’s Chemical Structure
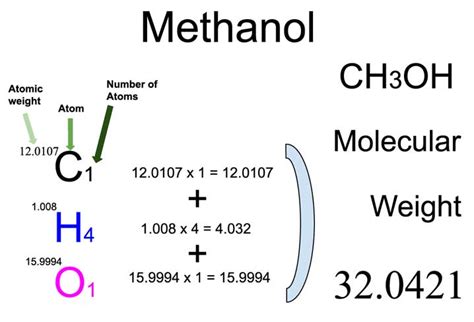
Methanol’s chemical structure comprises one carbon atom, four hydrogen atoms, and one oxygen atom, represented by the chemical formula CH3OH. Each component’s atomic mass plays a critical role in determining the total molar mass. Carbon, having an atomic mass of approximately 12.01 g/mol, forms the backbone of methanol’s molecular weight. The hydrogen atoms contribute about 1.008 g/mol each, and the oxygen atom adds roughly 16.00 g/mol. The simple summation of these individual atomic weights results in the precise molar mass of methanol: 32.04 g/mol. This understanding is vital for any chemical equation that requires accurate measurements.The Calculation Process
To unveil the secret of methanol’s molar mass, we need to delve deeper into the calculation process. Here’s how to determine it step-by-step:- Identify the molecular composition: Methanol (CH3OH) consists of 1 carbon (C), 4 hydrogen (H) atoms, and 1 oxygen (O) atom.
- Sum the atomic masses: Use periodic table values to add the atomic masses together:
- Carbon (C): 12.01 g/mol
- Hydrogen (H): 4 x 1.008 g/mol = 4.032 g/mol
- Oxygen (O): 16.00 g/mol
- Total molar mass: Add these values to find the molar mass of methanol:
- 12.01 g/mol (C) + 4.032 g/mol (H) + 16.00 g/mol (O) = 32.04 g/mol
Accurate calculations of molar mass are critical in chemistry to ensure precise formulations and reactions, underscoring the practical relevance of knowing methanol’s molar mass.
How does molar mass affect chemical reactions?
The molar mass of methanol is integral in determining the proportion of reactants needed for a balanced chemical reaction. It allows chemists to calculate how much of each reactant is required to produce a desired amount of product.
What happens if the molar mass is inaccurate?
Inaccurate molar mass calculations can lead to incorrect stoichiometric ratios, potentially causing failed experiments, improper product yields, and inefficiencies in industrial processes.
In conclusion, methanol’s molar mass isn’t just a number; it’s a cornerstone for precise chemical calculations and practical applications across various industries. Understanding and correctly applying this information ensures that chemical processes are executed with accuracy and efficiency.