In the realm of chemistry, understanding the molar mass of various compounds is foundational for a host of applications, from stoichiometric calculations in lab settings to theoretical chemical research. Specifically, C3H8, or propane, stands out as a staple for various practical purposes, including as fuel in domestic heating systems and in various industrial processes. This guide will provide a step-by-step, actionable approach to grasp and calculate the molar mass of C3H8, offering real-world examples and problem-solving strategies to tackle common user challenges.
The Need for Understanding Molar Mass of C3H8
Knowing the molar mass of C3H8 (propane) is critical for both educational and practical reasons. When performing chemical reactions, especially in professional settings like laboratories and industries, calculating the exact amount of substances is crucial. Incorrect molar mass calculations can lead to significant errors in measurements, resulting in flawed experiments or even hazardous situations. Hence, getting a firm grasp of how to determine the molar mass of propane is essential.A common pitfall for beginners is confusion over the concept of molar mass itself. The molar mass, often expressed in grams per mole (g/mol), is the total mass of one mole of a given substance. Understanding this concept clears the path for accurate calculations.
Quick Reference
- Immediate action item: Identify the constituent atoms in propane (C3H8).
- Essential tip: Use the periodic table to find atomic masses for carbon (C) and hydrogen (H).
- Common mistake to avoid: Confusing atomic mass with molecular mass—remember it’s the sum of the atomic masses of all atoms in the molecule.
How to Calculate the Molar Mass of C3H8
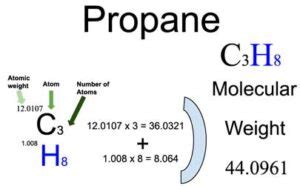
To calculate the molar mass of C3H8, we must sum the atomic masses of all constituent atoms. The steps are straightforward, but attention to detail is vital. Here’s a detailed how-to guide to ensure you get it right:Step 1: Determine the Atomic Mass of Each Element
Using the periodic table, note the atomic masses of carbon © and hydrogen (H). As of the latest updates:
- Carbon ©: 12.01 g/mol
- Hydrogen (H): 1.01 g/mol
Step 2: Identify the Number of Each Atom in the Molecule
For C3H8, the subscript numbers indicate the quantity of each type of atom:
- Carbon ©: 3 atoms
- Hydrogen (H): 8 atoms
Step 3: Calculate the Contribution of Each Element
Multiply the atomic mass of each element by the number of atoms present:
| Element | Atomic Mass | Quantity | Total Contribution |
|---|---|---|---|
| Carbon © | 12.01 g/mol | 3 | 3 x 12.01 g/mol = 36.03 g/mol |
| Hydrogen (H) | 1.01 g/mol | 8 | 8 x 1.01 g/mol = 8.08 g/mol |
Step 4: Add Up the Contributions to Find the Molar Mass
Sum the contributions from all the atoms:
36.03 g/mol (for carbon) + 8.08 g/mol (for hydrogen) = 44.11 g/mol
Final Answer
The molar mass of propane (C3H8) is 44.11 grams per mole.
Practical Examples to Implement
Now, let’s apply the calculated molar mass in practical scenarios.
Example 1: Preparing a Solution in the Lab
Suppose you need to prepare a solution that requires 2 moles of propane. Knowing its molar mass allows you to calculate the exact amount of propane to weigh out:
2 moles x 44.11 g/mol = 88.22 grams of propane.
This precise calculation ensures accurate formulation, critical for the integrity of the experiment.
Example 2: Calculating Fuel Requirements
In industrial applications, understanding propane’s molar mass helps in calculating fuel requirements for processes:
If a process requires 10 moles of propane, calculate:
10 moles x 44.11 g/mol = 441.1 grams of propane.
Accurate fuel calculation prevents wastage and ensures the process runs smoothly.
Common Pitfalls and How to Avoid Them
Understanding the molar mass of C3H8 is essential but comes with some challenges.
Avoiding Calculation Mistakes
One frequent error is miscounting the number of atoms or incorrect use of atomic masses. Always double-check:
- The number of each atom specified in the molecular formula.
- The accurate atomic mass from the latest periodic table.
Simple mistakes can lead to significant discrepancies, so verification is key.
Misunderstanding Molecular Mass vs. Molar Mass
Some learners confuse molecular mass with molar mass. Here’s the distinction:
- Molecular Mass: Sum of the atomic masses of atoms in a molecule.
- Molar Mass: This is the molecular mass in grams per mole.
Proper comprehension prevents confusion, especially in complex chemical calculations.
Can molar mass vary for different isotopes?
Typically, molar mass calculations use standard atomic masses from the periodic table, assuming a uniform isotopic composition of elements. For highly precise work, isotopic variations might matter, but for general purposes, this approach works perfectly fine.
What’s the significance of knowing the molar mass?
Knowing the molar mass is crucial for accurate stoichiometric calculations, ensuring precise amounts of reactants and products in chemical reactions. This precision is fundamental for successful lab experiments and industrial processes.
How can I easily remember the molar mass of common compounds?
Memorizing can be overwhelming, but it helps to break it down. Use periodic table references and memorize common patterns and rules. For instance, water (H2O) has a molar mass of 18.01 g/mol (2 x 1.01 g/mol for H + 16.00 g/mol for O). Practice with a few more to build your familiarity.
Through this guide, you now have a robust framework to calculate the molar mass of C3H8. By following the steps, utilizing practical examples, and avoiding common pitfalls, you’ll master this fundamental skill in chemistry with confidence and precision.