Are you struggling to understand the molar mass of barium hydroxide, or are you finding it challenging to calculate it accurately? You’re not alone in facing this chemistry conundrum. Determining the molar mass of compounds like barium hydroxide can be daunting but is a fundamental skill necessary for both students and professionals in the scientific field. This guide will break down the molar mass calculation step-by-step, with real-world examples and practical solutions to help you master this essential chemistry concept. Whether you are preparing for an exam, conducting a lab experiment, or simply deepening your scientific knowledge, this detailed and actionable guide will provide you with the precise information you need.
Understanding the Molar Mass of Barium Hydroxide
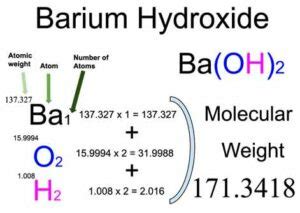
The molar mass of a compound is the sum of the atomic masses of all atoms in its molecular formula. Barium hydroxide, with the chemical formula Ba(OH)₂, consists of one barium atom (Ba), two oxygen atoms (O), and two hydrogen atoms (H). To find the molar mass, we need to sum up the atomic masses of these constituent atoms. This process is vital for understanding chemical reactions, stoichiometry, and other critical aspects of chemistry.
Problem-Solution Opening Addressing User Needs
Deciphering the molar mass might seem like a complex puzzle, especially when dealing with compounds containing multiple elements. However, with a structured approach, it becomes straightforward. If you're consistently confused by the numbers and want to avoid common mistakes, this guide will walk you through each step, providing not only precise calculations but also practical tips and examples. We will address the most common pain points, such as ensuring accuracy in atomic mass values and avoiding calculation errors. By the end of this guide, you'll have a clear, actionable strategy for calculating the molar mass of barium hydroxide and other compounds.
Quick Reference
Quick Reference
- Immediate action item: Identify the atomic masses of all elements in barium hydroxide.
- Essential tip: Multiply the atomic mass of each element by its quantity in the formula and then sum these values.
- Common mistake to avoid: Forgetting to account for the correct subscripts in the chemical formula.
Detailed How-To Sections
Calculating the Molar Mass of Barium Hydroxide
To calculate the molar mass of barium hydroxide, follow these steps:
Step-by-Step Calculation
1. Identify the atomic masses of each element in the compound.
- Barium (Ba): The atomic mass of barium is approximately 137.33 g/mol.
- Oxygen (O): The atomic mass of oxygen is approximately 16.00 g/mol.
- Hydrogen (H): The atomic mass of hydrogen is approximately 1.01 g/mol.
2. Determine the quantity of each element in the molecular formula.
Barium Hydroxide has the formula Ba(OH)₂, indicating:
- 1 barium atom
- 2 oxygen atoms
- 2 hydrogen atoms
3. Calculate the total mass contributed by each element.
- For barium: 1 * 137.33 g/mol = 137.33 g/mol
- For oxygen: 2 * 16.00 g/mol = 32.00 g/mol
- For hydrogen: 2 * 1.01 g/mol = 2.02 g/mol
4. Sum the masses to get the total molar mass.
Total molar mass of Ba(OH)₂ = 137.33 g/mol + 32.00 g/mol + 2.02 g/mol = 171.35 g/mol
Practical Example
Let’s apply this calculation to a practical scenario. Suppose you are tasked with preparing a 0.100 mol solution of barium hydroxide for a lab experiment. To determine the mass of barium hydroxide needed, you need to know its molar mass:
- Molar mass of Ba(OH)₂ = 171.35 g/mol
To prepare 0.100 mol of Ba(OH)₂, you need:
- Mass = moles × molar mass
- Mass = 0.100 mol × 171.35 g/mol
- Mass = 17.135 grams
Thus, you need 17.135 grams of barium hydroxide to prepare 0.100 mol of the solution.
Common Mistakes to Avoid
Even with careful calculation, mistakes can occur. Here are some common errors and how to avoid them:
- Mistake: Using outdated atomic mass values.
- Mistake: Ignoring subscripts in the chemical formula.
- Mistake: Adding rather than multiplying when calculating the mass of an element.
Solution: Always use the most recent atomic mass values from reliable scientific sources like the Periodic Table.
Solution: Ensure you correctly interpret the subscripts, which tell you how many atoms of each element are present.
Solution: For each element, multiply its atomic mass by the number indicated by its subscript in the chemical formula.
Advanced Considerations
For those looking to delve deeper, consider these advanced concepts:
Using Molar Mass in Stoichiometry
Understanding molar mass is crucial in stoichiometry, where you determine the quantities of reactants and products in chemical reactions. For example, if you have a balanced chemical equation and know the molar mass, you can calculate how much of each reactant to use or how much product to expect.
Empirical vs. Molecular Formula
Sometimes, you might encounter empirical formulas, which represent the simplest whole-number ratio of elements in a compound. To convert an empirical formula to a molecular formula, you need to know the molar mass of the compound and compare it with the empirical formula’s molar mass. For barium hydroxide, if the empirical formula is BaOH and you know the molar mass, you can determine that Ba(OH)₂ is the molecular formula.
Practical FAQ
How do I find the atomic masses for elements in the compound?
Atomic masses are found on the Periodic Table of Elements. They are usually listed in atomic mass units (amu) but are often converted to grams per mole (g/mol) for practical lab calculations. Always use the most recent values from a reliable source to ensure accuracy.
What should I do if my calculated molar mass doesn’t match the literature value?
If your calculated molar mass differs from the literature value, recheck your steps:
- Verify atomic masses.
- Ensure correct interpretation of subscripts in the chemical formula.
- Double-check multiplication for the correct quantities of each element.
- Consider rounding errors.
Small errors in any of these steps can lead to discrepancies.
Can I use an online calculator for molar mass instead of manual calculations?
While online calculators are handy, understanding manual calculations provides deeper insight into the underlying principles of chemistry. However, if you need a quick verification or are working on larger, more complex compounds, online tools can be efficient. Ensure the calculator uses current atomic mass values.