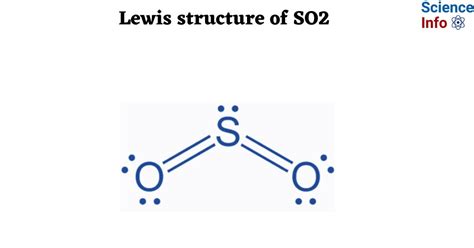
Understanding SO2’s Lewis Structure: Key Insights Unveiled
If you’re here, you’re likely tackling the complexities of sulfur dioxide’s (SO₂) Lewis structure and wondering how to draw it effectively. SO₂ is a fundamental molecule that plays a pivotal role in environmental chemistry, atmospheric science, and industrial applications. This guide will walk you through every aspect you need to understand its Lewis structure, from fundamental principles to advanced insights.
Let’s dive right in to get you started on the path to mastering SO₂’s Lewis structure.
Problem-Solution Opening Addressing User Needs
Understanding the Lewis structure of SO₂ is crucial for students and professionals in chemistry, as it offers essential insights into the molecular geometry, bonding, and potential reactivity of the compound. This can significantly impact fields such as environmental science and industrial processes, where SO₂ acts as a precursor to sulfuric acid and a pollutant in fossil fuel combustion. However, the challenge often lies in interpreting valence electrons, understanding the octet rule, and grasping the nuances of resonance structures. This guide is designed to demystify these complexities by offering step-by-step, actionable advice, complete with practical examples and real-world applications.
Quick Reference
Quick Reference
- Immediate action item: Count the valence electrons of sulfur and oxygen to start.
- Essential tip: Place the least electronegative atom (sulfur) in the center and surround it with the more electronegative oxygens.
- Common mistake to avoid: Failing to distribute the remaining electrons to satisfy the octet rule, particularly around oxygen.
Step-by-Step Guide to Drawing SO₂'s Lewis Structure
Drawing the Lewis structure for SO₂ may seem daunting at first, but by breaking it down into clear, manageable steps, you can simplify the process. Follow these comprehensive instructions to master the structure.
Step 1: Determine the Total Number of Valence Electrons
To begin, identify the valence electrons for sulfur and oxygen:
- Sulfur (S) has 6 valence electrons.
- Each oxygen (O) has 6 valence electrons.
SO₂ consists of one sulfur atom and two oxygen atoms. Therefore, the total number of valence electrons is:
Total valence electrons = 6 (S) + 2 * 6 (O) = 18 valence electrons
Step 2: Place the Least Electronegative Atom in the Center
In SO₂, sulfur is less electronegative than oxygen. Place sulfur in the center of the structure:
O —— S —— O
Draw a single bond from each oxygen to sulfur:
O ——S—— O
Step 4: Distribute the Remaining Electrons to Satisfy the Octet Rule
Begin by placing the remaining electrons around the outer atoms (oxygens) to satisfy their octets. Each oxygen atom needs 8 electrons:
- Two electrons are already part of the single bond (one from each oxygen and sulfur). Each oxygen atom has two more electrons to get to an octet.
Distribute the remaining 12 electrons (6 pairs) as follows:
O ——S—— O
You’ll now place two lone pairs on each oxygen:
O ——S—— O
This uses up 8 electrons on each oxygen, leaving 4 electrons to be distributed.
Step 5: Complete the Octet on the Central Atom
Sulfur now needs 2 more electrons to complete its octet (since it has four single bonds already). Distribute the remaining 4 electrons to sulfur:
Lone pairs on Oxygens: O (: : ) ——S—— (: : ) O
This leaves sulfur with two lone pairs (4 electrons), completing its octet.
Step 6: Check Resonance Structures
SO₂ can exhibit resonance, meaning the electrons can be distributed differently while maintaining the same skeleton. In SO₂, there are two possible resonance structures:
Each oxygen atom can double bond to sulfur:
O =S—O
And,
O =S——O
While these structures are theoretical, they indicate that the true structure is an average of the two, with each S—O bond being somewhere between a single and double bond in length.
Step 7: Confirm the Lewis Structure
Review your structure:
- Each oxygen atom has an octet (8 electrons).
- Sulfur has an expanded octet with 10 electrons.
- The total number of valence electrons used is 18.
This structure fulfills all conditions for a Lewis structure.
Practical FAQ
How can I identify potential mistakes in drawing SO₂'s Lewis structure?
Common errors include not distributing electrons correctly to satisfy the octet rule, misplacing the least electronegative atom, and failing to recognize resonance structures. When you encounter these issues, revisit the step of calculating valence electrons and ensure each atom achieves a complete octet (or duet for hydrogen). Double-check your final structure to confirm that the total valence electrons are accounted for.
Why does sulfur have an expanded octet in SO₂?
Sulfur is in group 6A and has five d-orbitals in addition to its sp3 hybrid orbitals. In SO₂, sulfur forms four sigma bonds and has two lone pairs. The ability of sulfur to utilize its d-orbitals allows it to accommodate more than eight electrons, resulting in an expanded octet.
What does the presence of resonance structures imply?
Resonance structures indicate that the actual electronic structure of a molecule is a hybrid of the possible Lewis structures. For SO₂, this means the true structure is an average of the resonance forms, with partial double bond character in each S—O bond. This leads to a bond length between a single and double bond, which is shorter than in typical single bonds but longer than in typical double bonds.
By adhering to these guidelines, you’ll be able to draw SO₂’s Lewis structure confidently and understand its fundamental chemical properties. This comprehensive, user-focused approach will help you gain the knowledge and practical skills needed to tackle similar molecules in the future. Happy studying!