Determining the molar mass of compounds is a fundamental skill in chemistry that provides crucial information for a wide range of applications, from laboratory experiments to industrial processes. For example, potassium sulfate (K2SO4), a vital compound used in fertilizers, has a molar mass that needs to be accurately calculated to understand its chemical properties and practical uses. In this guide, we’ll walk you through step-by-step instructions on how to determine the molar mass of K2SO4, along with practical tips, common pitfalls, and frequently asked questions. Let’s dive in!
Understanding the Need for Molar Mass Calculation
The molar mass of a compound is the mass of one mole of that substance, typically expressed in grams per mole (g/mol). Calculating the molar mass of K2SO4 is essential for several reasons:
- To ensure accurate stoichiometric calculations in chemical reactions.
- To determine the correct quantities needed for laboratory experiments.
- To understand the material’s properties and its usage in various fields such as agriculture and manufacturing.
Knowing how to determine the molar mass of K2SO4 will empower you to carry out precise and effective chemical analyses.
Quick Reference Guide
Quick Reference
- Immediate action item with clear benefit: Use the periodic table to find atomic masses for each element in K2SO4.
- Essential tip with step-by-step guidance: Add the individual masses together, keeping in mind the number of each type of atom in the formula.
- Common mistake to avoid with solution: Don’t forget to multiply the atomic mass of each element by the number of its atoms in the compound.
Calculating the Molar Mass of K2SO4
To determine the molar mass of K2SO4, we need to understand the composition of the compound and sum the atomic masses of its constituent elements. Here’s how you can do it:
Step-by-Step Guide to Calculating Molar Mass
Follow these detailed steps to calculate the molar mass of K2SO4 effectively:
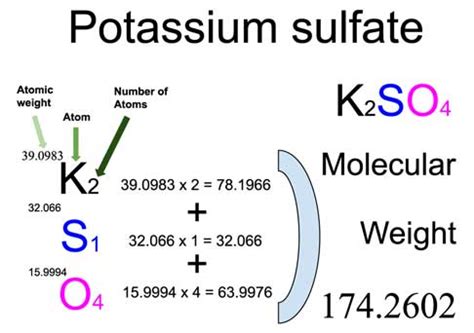
- Identify the Elements and Their Quantities: K2SO4 consists of potassium (K), sulfur (S), and oxygen (O). There are 2 potassium atoms, 1 sulfur atom, and 4 oxygen atoms in each molecule of K2SO4.
- Find Atomic Masses: Use the periodic table to find the atomic mass of each element:
Element Atomic Mass (g/mol) K (Potassium) 39.10 S (Sulfur) 32.07 O (Oxygen) 16.00 - Calculate Individual Contributions: Multiply each atomic mass by the number of atoms of that element in the compound:
- For Potassium: 39.10 g/mol * 2 = 78.20 g/mol
- For Sulfur: 32.07 g/mol * 1 = 32.07 g/mol
- For Oxygen: 16.00 g/mol * 4 = 64.00 g/mol
- Sum the Contributions: Add up all the contributions to get the total molar mass of K2SO4:
- Total Molar Mass = 78.20 g/mol (K) + 32.07 g/mol (S) + 64.00 g/mol (O) = 174.27 g/mol
Therefore, the molar mass of K2SO4 is 174.27 g/mol.
Practical Examples and Applications
Let’s explore some practical examples to understand how knowing the molar mass of K2SO4 can be beneficial in real-world scenarios.
Example 1: Preparing a Solution
Imagine you need to prepare a 0.5 M solution of K2SO4 in a 1-liter volume for a laboratory experiment. Here's how you would calculate the required mass:
- Determine moles needed: For a 0.5 M solution in 1 liter, you need 0.5 moles of K2SO4.
- Calculate mass: Using the molar mass of K2SO4 (174.27 g/mol), you multiply by the number of moles: 0.5 moles * 174.27 g/mol = 87.14 grams
- Prepare the solution: Weigh out 87.14 grams of K2SO4 and dissolve it in 1 liter of distilled water.
By knowing the molar mass, you ensure that your solution is prepared accurately, which is crucial for the success of your experiment.
Example 2: Fertilizer Composition
In agriculture, K2SO4 is used as a potassium fertilizer. To determine the percentage of potassium in the fertilizer, we first find the mass of potassium in one mole of K2SO4:
- Calculate potassium mass: Each K2SO4 molecule contains 2 potassium atoms, with an atomic mass of 39.10 g/mol each, so: 2 * 39.10 g/mol = 78.20 g/mol
- Calculate the percentage: To find the percentage of potassium by mass in K2SO4, divide the mass of potassium by the total molar mass and multiply by 100: (78.20 g/mol / 174.27 g/mol) * 100 ≈ 45%
Thus, K2SO4 is approximately 45% potassium, which is important information for determining the nutrient content of the fertilizer.
Frequently Asked Questions (FAQ)
What is the molar mass of K2SO4?
The molar mass of K2SO4 is calculated by adding the atomic masses of its constituent elements, taking into account the number of each type of atom in the formula. As detailed in the previous sections, the total molar mass is 174.27 g/mol.