Water, a fundamental component of life, has puzzled scientists for centuries. Its unique molecular structure raises intriguing questions about its polarity. This article explores the question, “Is water nonpolar?” delving into molecular mysteries and providing expert insights to clear up any confusion.
Key insights box:
Key Insights
- Water (H₂O) is a polar molecule due to its bent shape and electronegativity differences.
- The molecular structure leads to uneven charge distribution, creating a dipole moment.
- Understanding water’s polarity is crucial for grasping its role in chemical and biological processes.
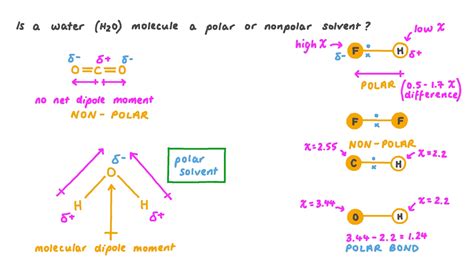
The debate over water’s polarity often hinges on misunderstanding its molecular makeup. Water consists of two hydrogen atoms covalently bonded to a single oxygen atom. This arrangement forms a bent molecular geometry, with the hydrogen atoms positioned 104.5 degrees apart from each other. The oxygen atom’s higher electronegativity compared to hydrogen atoms results in the oxygen atom drawing shared electrons closer to itself.
This uneven electron distribution creates regions with partial charges. The oxygen atom bears a partial negative charge, while each hydrogen atom has a partial positive charge. Consequently, water possesses a net dipole moment, indicating it is a polar molecule.
Analysis of water’s molecular structure The molecular geometry of water is pivotal in understanding its polarity. The electron-pair repulsion theory (VSEPR theory) explains that the two lone pairs of electrons on the oxygen atom, along with the two bonding pairs to the hydrogen atoms, occupy positions that minimize repulsion. This results in a bent molecular shape with an angle of 104.5 degrees. The bent structure ensures that the positive and negative charges do not cancel each other out, maintaining a net dipole moment.
In practical terms, the polarity of water has far-reaching implications. It explains why water molecules are strongly attracted to each other, forming hydrogen bonds. This characteristic is responsible for water’s high surface tension and its ability to dissolve many substances—both ionic and polar compounds—through a process known as solvation. These properties are fundamental in various natural processes, from cloud formation to nutrient transport in biological systems.
Technical consideration with clear application A technical consideration pertinent to the polarity of water is the concept of electronegativity. Oxygen’s electronegativity value of 3.44, compared to hydrogen’s 2.20, leads to an uneven electron distribution. This disparity means that the electrons in the covalent bonds between oxygen and hydrogen spend more time around the oxygen atom, making the oxygen atom partially negative and the hydrogen atoms partially positive.
Understanding this technical aspect is essential for fields such as chemistry and biochemistry. For example, the polarity of water is crucial in understanding enzyme-substrate interactions in biological systems. The hydrogen bonds formed between water molecules and various substrates can either facilitate or inhibit enzymatic reactions, impacting metabolic pathways significantly.
Actionable recommendation To leverage the understanding of water’s polarity in practical applications, researchers and scientists should focus on harnessing its unique solvent properties in drug design and development. Water’s ability to dissolve ionic and polar molecules makes it an excellent medium for various biochemical reactions and processes. Thus, considering water’s polarity when designing new molecules or compounds can significantly enhance solubility and bioavailability, crucial for effective therapeutic agents.
FAQ section:
What role does water’s polarity play in chemical reactions?
Water’s polarity allows it to act as an excellent solvent for ionic and polar substances. This property is crucial in facilitating many chemical reactions in biological systems, where water often serves as the reaction medium. The polar nature of water helps to stabilize charges and enables the formation of hydrogen bonds, which are fundamental in various biochemical processes.
How does the polarity of water affect its physical properties?
The polarity of water significantly influences its physical properties such as surface tension, boiling and melting points, and thermal capacity. The hydrogen bonds between water molecules create a strong intermolecular force, leading to higher surface tension compared to nonpolar liquids. This high surface tension allows small insects to walk on water. Additionally, water’s polarity results in higher boiling and melting points compared to other molecules of similar size, due to the energy required to break hydrogen bonds.
Understanding whether water is nonpolar or polar not only satisfies scientific curiosity but also has practical implications in numerous scientific disciplines. Through this detailed examination, we’ve clarified water’s inherent polarity and underscored its critical role in molecular and biological systems.