Understanding how to calculate mass from density and volume is fundamental in various scientific and engineering disciplines. Density, the measure of mass per unit volume, provides a crucial parameter for various applications, ranging from material science to environmental studies. This article delves into the relationship between mass, density, and volume, providing a clear, practical guide for accurate calculations.
Fundamental Concepts: Density, Mass, and Volume
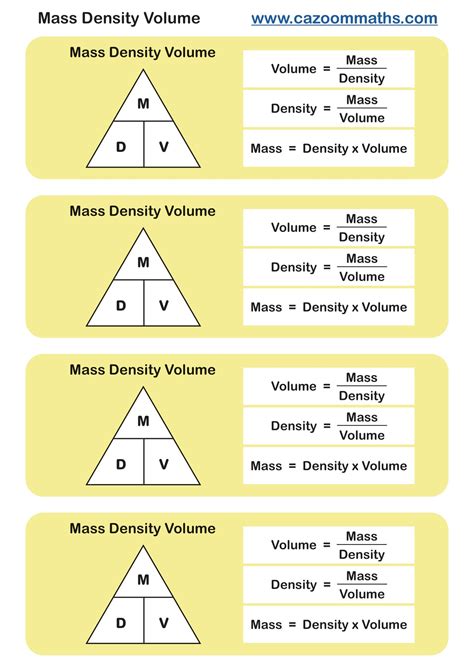
Density (ρ) is defined as mass (m) per unit volume (V), represented by the formula: ρ = m/V. This simple equation is central to numerous scientific calculations. To determine mass from density and volume, rearrange the formula to m = ρ * V. This relationship highlights the necessity of precise measurements for density and volume to ensure accuracy in mass calculations.
Why Density Matters
Density is a critical property that helps in identifying substances. For instance, lead has a density of approximately 11.34 g/cm³, while aluminum has a lower density of 2.70 g/cm³. Knowing these values allows scientists and engineers to select materials based on their density for specific applications. Accurate density and volume measurements are essential for calculating mass, ensuring that materials meet required specifications.
Step-by-Step Calculation
To calculate mass from density and volume, follow these steps:
- Measure the density (ρ) of the substance in the appropriate unit, such as grams per cubic centimeter (g/cm³) or kilograms per cubic meter (kg/m³).
- Determine the volume (V) of the substance in compatible units, like cubic centimeters (cm³) or cubic meters (m³).
- Use the formula m = ρ * V to calculate mass. Ensure all units are consistent for accuracy.
For example, consider a cube of aluminum with a side length of 10 cm. First, calculate the volume: V = 10 cm * 10 cm * 10 cm = 1000 cm³. Given the density of aluminum is 2.7 g/cm³, the mass calculation would be: m = 2.7 g/cm³ * 1000 cm³ = 2700 grams.
Key Insights
- Primary insight with practical relevance: Accurate mass calculations depend on precise density and volume measurements, essential for material selection and application.
- Technical consideration with clear application: Understanding the relationship between mass, density, and volume helps in fields like material science for selecting optimal materials.
- Actionable recommendation: Always ensure unit consistency when calculating mass from density and volume to avoid errors.
Real-World Applications
Calculating mass from density and volume is essential in many real-world scenarios. In aerospace engineering, precise calculations ensure that aircraft components meet strict weight requirements. In chemistry, determining the mass of reactants and products is crucial for understanding reaction dynamics. Additionally, environmental scientists use these calculations to estimate pollutant loads in water bodies or soil samples.
FAQ Section
What happens if I have the density in kg/m³ and volume in cm³?
To maintain unit consistency, convert volume to cubic meters. 1 cm³ equals 1 x 10⁻⁶ m³. Thus, for a volume of 1000 cm³, it is 0.001 m³.
Can density change for a given substance?
Density can change with temperature and pressure. For most practical applications, density is considered constant at standard conditions.
Understanding how to calculate mass from density and volume is vital for various scientific and engineering fields. Accurate calculations hinge on precise measurements and maintaining consistent units. By grasping these fundamental principles, professionals can ensure their work meets the required standards, ultimately contributing to advancements in technology, safety, and environmental conservation.