Unlock Atomic Mass Secrets Today!
If you’ve ever found yourself baffled by the complexities of atomic mass, you’re not alone. Understanding atomic mass can be challenging, especially when dealing with the often confusing world of subatomic particles. However, once you grasp the fundamental concepts, the information opens up a deeper understanding of chemistry and physics. This guide is designed to demystify atomic mass, providing you with the actionable advice and practical solutions to help you master this topic.
Problem-Solution Opening Addressing User Needs
The enigmatic world of atomic mass can often leave learners and even seasoned scientists scratching their heads. This isn't just a theoretical hurdle; it has practical implications for fields ranging from chemistry to materials science and even nuclear physics. Many struggle to understand how to calculate atomic mass, what role it plays in chemical reactions, and how isotopic variations impact these calculations. This guide is tailored to provide step-by-step guidance with actionable advice. We'll take you through real-world examples and practical solutions, making the complex concepts of atomic mass accessible and straightforward.
By the end of this guide, you’ll not only understand atomic mass but also how to apply this knowledge in various practical scenarios, from laboratory settings to real-world industrial processes. Let's dive in and unlock the secrets of atomic mass together!
Quick Reference
Quick Reference
- Immediate action item: To get the hang of atomic mass, start with the most common isotope of each element. This will make it easier to build your understanding.
- Essential tip: Use the periodic table to find the atomic mass number; it’s usually found below the symbol of the element. This number represents the weighted average mass of all the isotopes of an element.
- Common mistake to avoid: Confusing atomic mass with molecular mass. Remember, atomic mass pertains to single atoms, while molecular mass concerns the entire compound.
Detailed How-To Sections
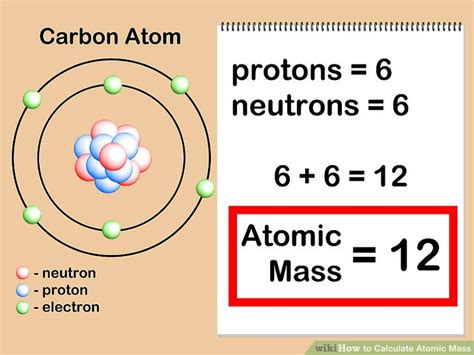
Understanding Atomic Mass
Atomic mass, also known as atomic weight, is a crucial concept that combines the number of protons and neutrons in an atom’s nucleus. Here’s a step-by-step guide to comprehending atomic mass:
- Step 1: Find the atomic number on the periodic table, which tells you the number of protons in the nucleus of an atom.
- Step 2: Determine the most common isotope of the element to get the most straightforward introduction. For instance, the most common isotope of carbon is Carbon-12.
- Step 3: Locate the atomic mass number on the periodic table, usually found below the element’s symbol.
- Step 4: Understand that the atomic mass is the weighted average of all isotopes of an element. For Carbon-12, the atomic mass is approximately 12 amu (atomic mass units).
- Step 5: Realize that isotopes are variants of an element that have the same number of protons but different numbers of neutrons. The atomic mass is calculated by averaging the masses of all isotopes, weighted by their natural abundance.
Let’s look at an example. For Carbon-12, the atomic mass is 12 amu. This is the sum of its 6 protons and 6 neutrons. Carbon-13 and Carbon-14 are isotopes that occur less frequently but contribute to the atomic mass calculation through their natural abundance.
Calculating Atomic Mass for Various Isotopes
To calculate the atomic mass for elements that have multiple isotopes, you need to consider the natural abundance of each isotope:
- Step 1: Identify the isotopes and their respective abundances. For instance, Chlorine has two major isotopes: Chlorine-35 and Chlorine-37. Chlorine-35 constitutes about 75.78% of natural chlorine, while Chlorine-37 makes up about 24.22%.
- Step 2: Convert the percentages into decimal form. 75.78% becomes 0.7578 and 24.22% becomes 0.2422.
- Step 3: Multiply the mass of each isotope by its respective decimal abundance. For Chlorine-35, this would be 35 * 0.7578, and for Chlorine-37, it would be 37 * 0.2422.
- Step 4: Add these values together to get the average atomic mass. For Chlorine, this would be (35 * 0.7578) + (37 * 0.2422) = 26.98 + 8.97 = 35.95 amu.
This method ensures that you account for the natural occurrence of each isotope, leading to a more accurate atomic mass calculation.
Applying Atomic Mass in Chemical Reactions
Understanding atomic mass isn’t just for theoretical knowledge—it’s also essential for practical applications in chemical reactions. Here’s how to use atomic mass effectively:
- Step 1: Determine the molecular formula of the compound. For example, consider water (H2O).
- Step 2: Identify the atomic masses of each element involved. Hydrogen has an atomic mass of about 1 amu, and Oxygen has an atomic mass of about 16 amu.
- Step 3: Multiply the atomic mass of each element by the number of atoms of that element in the molecule. For water, this would be (2 * 1 amu for Hydrogen) + (1 * 16 amu for Oxygen).
- Step 4: Sum these values to find the molecular mass of the compound. For water, it would be (2 * 1) + (1 * 16) = 18 amu.
This step-by-step approach provides a practical understanding of how atomic mass is applied in real-world chemical reactions.
Practical FAQ
Why is atomic mass important in chemistry?
Atomic mass is crucial for understanding and predicting the behavior of atoms and molecules in chemical reactions. It helps in determining the molar mass of compounds, which is fundamental for calculations involving stoichiometry. Knowing the atomic mass allows scientists to accurately predict the mass ratios in which elements combine to form compounds. This knowledge is essential for a wide range of applications, from synthesizing new materials to determining the composition of unknown substances.
How does isotopic variation affect atomic mass?
Isotopes are variants of a chemical element that have different numbers of neutrons and thus different atomic masses. The atomic mass you find on the periodic table is the weighted average of all naturally occurring isotopes of an element. For example, carbon has three isotopes: Carbon-12, Carbon-13, and Carbon-14. Carbon-12 is the most abundant isotope, making up about 98.9% of carbon, whereas Carbon-13 and Carbon-14 are much rarer. The atomic mass listed is calculated based on the natural abundance of these isotopes, providing an average mass that represents the element in nature.
Can atomic mass vary for the same element?
Yes, atomic mass can vary for the same element due to the presence of different isotopes. Each isotope has a different mass because it has a different number of neutrons. While the atomic number (which defines the element) remains constant, the atomic mass will differ depending on the isotope. For example, chlorine has two stable isotopes, Chlorine-35 and Chlorine-37, which lead to the atomic mass of chlorine being an average of the masses of these isotopes, weighted by their natural abundances.