Mastering the HCN Lewis Structure: The Ultimate Guide for Chemistry Enthusiasts
Understanding the HCN Lewis structure is a fundamental aspect of mastering organic chemistry. The structure of HCN, or hydrogen cyanide, plays a vital role in determining its properties and reactions. This guide aims to walk you through each step of creating the Lewis structure for HCN, addressing common challenges, and providing actionable advice to ensure a thorough understanding.
Introduction to HCN Lewis Structure
Hydrogen cyanide (HCN) is a simple yet fascinating molecule that serves as a cornerstone in the study of molecular bonding and structure. The Lewis structure of HCN allows us to visualize the arrangement of atoms and electrons, offering insight into its chemical behavior. This guide will provide you with a comprehensive overview, ensuring you grasp both the basics and complexities involved in constructing an accurate Lewis structure for HCN.
Problem-Solution Opening
Creating an accurate Lewis structure for HCN can initially seem challenging due to the intricacies of electron distribution and molecular geometry. Many students struggle with identifying the central atom, determining the valence electrons, and ensuring the structure follows the octet rule. Our goal in this guide is to simplify the process step-by-step, providing clear, actionable advice to overcome these challenges. By the end of this guide, you’ll have the knowledge to not only construct a Lewis structure for HCN but also to understand its underlying chemical principles.
Quick Reference
Quick Reference
- Immediate action item: Determine the valence electrons for each atom. Hydrogen has 1, carbon has 4, and nitrogen has 5.
- Essential tip: Always place the least electronegative atom in the center of the structure. In HCN, carbon is the central atom.
- Common mistake to avoid: Miscalculating the total valence electrons. Ensure you sum the electrons correctly before proceeding.
Detailed Steps to Create an HCN Lewis Structure
To construct an accurate HCN Lewis structure, follow these detailed steps:
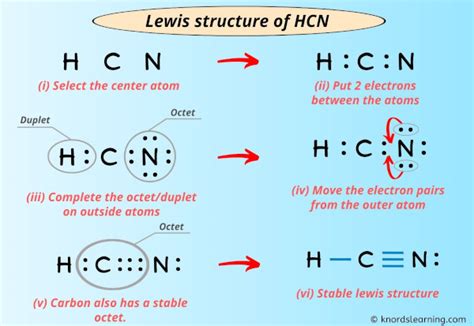
Step 1: Calculate Total Valence Electrons
The first step is to sum up the valence electrons from all atoms in the molecule.
- Hydrogen (H): 1 valence electron
- Carbon ©: 4 valence electrons
- Nitrogen (N): 5 valence electrons
Total valence electrons = 1 (H) + 4 © + 5 (N) = 10 valence electrons.
Step 2: Determine the Central Atom
The central atom is typically the least electronegative atom in the molecule. Here, carbon is less electronegative than hydrogen and nitrogen, so it will be the central atom.
Step 3: Arrange the Atoms
Place the carbon atom in the center, with the hydrogen atom bonded to one side and the nitrogen atom bonded to the other side. This gives the structure H—C≡N.
Step 4: Connect Atoms with Single Bonds
Start by connecting the central atom with single bonds to the surrounding atoms.
C—H and C—N.
Step 5: Distribute Remaining Electrons
Distribute the remaining valence electrons to satisfy the octet rule. Start with the atoms surrounding the central atom.
- Carbon needs 4 more electrons to complete its octet.
- Hydrogen already has 2 electrons (a full shell for H).
- Nitrogen needs 2 more electrons to complete its octet.
Place 2 lone pairs on the nitrogen atom.
Step 6: Verify the Octet Rule
Check that each atom in the structure has a complete octet (or duet for hydrogen).
Hydrogen: Full shell with 2 electrons.
Carbon: 8 electrons (4 from bonds + 4 from lone pairs).
Nitrogen: 8 electrons (3 from bonds + 2 lone pairs).
Step 7: Modify Structure if Necessary
If any atom does not have a complete octet, move electrons from other atoms to form multiple bonds if needed. In HCN, the structure already satisfies the octet rule.
Practical Examples
Let’s apply these steps to an example where HCN undergoes a chemical reaction:
When HCN is reacted with sodium cyanide (NaCN), the nitrogen atom in HCN usually ends up forming a triple bond with carbon and a single bond with hydrogen. This is a practical application of the Lewis structure concept in a real-world chemical reaction.
Practical FAQ
Why is the carbon atom chosen as the central atom in HCN?
The central atom in a molecule is typically the least electronegative atom to maximize its bonding capacity and central positioning in the molecule’s geometry. Carbon has an electronegativity value of 2.55, which is less than that of hydrogen (2.20) and nitrogen (3.04). By positioning carbon at the center, the molecule achieves greater stability as carbon can effectively distribute its valence electrons to satisfy its bonding needs.
How can I determine the correct number of bonds an atom can form?
To determine the number of bonds an atom can form, consider the number of valence electrons it has and the number of electrons needed to complete its octet (or duet for hydrogen). Carbon, for example, has 4 valence electrons and needs 4 more to complete its octet. It can form four single bonds, or one triple bond. Hydrogen can form one single bond, and nitrogen can form three single bonds or one triple bond. To figure out the correct number of bonds, look at the structure you're building and how the electrons can be shared to satisfy the octet rule for all atoms involved.
Best Practices and Tips
As you work on more complex molecules, consider these best practices:
- Practice Regularly: Frequent practice helps you quickly identify patterns and exceptions in electron distribution.
- Use Diagrams: Sketching the structure repeatedly aids in visual understanding and retention.
- Understand Molecular Geometry: Knowing the different shapes molecules can take (VSEPR theory) helps in predicting how atoms will arrange themselves.
- Check Resonance Structures: Some molecules have multiple valid Lewis structures due to resonance. Consider all possibilities to ensure you’re accounting for all bonding configurations.
- Double-Check Electron Counts: Carefully counting valence electrons at each step ensures no errors in your structure.
With these guidelines, you’ll be well on your way to mastering the HCN Lewis structure and beyond. Remember, the key is to practice, ask questions, and continually test your understanding through different examples and applications.