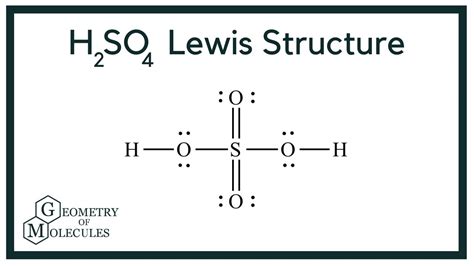
Understanding the H2SO4 Lewis structure is essential for anyone diving into advanced chemistry. This guide aims to demystify the structure, offering a step-by-step approach to grasp this important concept, filled with actionable advice, practical solutions, and expert insights to ensure you're equipped to master it.
H2SO4, commonly known as sulfuric acid, plays a crucial role in both industrial and laboratory settings. Grasping its Lewis structure not only helps in understanding its chemical behavior but also forms a foundation for learning about more complex molecules. Let’s dive into a practical guide that demystifies this complex structure, offering you real-world examples and actionable steps to ensure your success.
Immediate Action: Identify Sulfur as the Central Atom
When drawing the Lewis structure for H2SO4, start by identifying the central atom, which in this case is sulfur (S). Sulfur's ability to expand its octet due to its d-orbitals allows it to form multiple bonds, making it a perfect central atom for this molecule.
Quick Reference
- Immediate action item: Always place sulfur as the central atom when drawing the H2SO4 Lewis structure.
- Essential tip: Sulfur can expand its octet due to the availability of d-orbitals, allowing it to form more than eight electrons around it.
- Common mistake to avoid: Mistaking oxygen or hydrogen as the central atom, which does not allow for the proper bonding and resonance structures.
Drawing the H2SO4 Lewis Structure: Step-by-Step
Drawing the Lewis structure for H2SO4 is a straightforward yet meticulous process. Here, we’ll break it down into clear steps to ensure you understand each part:
Step 1: Count the total valence electrons. Sulfur has 6, each hydrogen atom has 1, and each oxygen atom has 6.
Step 2: Arrange the atoms. Place sulfur at the center and surround it with the two oxygen atoms and two hydrogen atoms.
Step 3: Form single bonds. Draw single bonds between sulfur and each oxygen atom, and single bonds between sulfur and each hydrogen atom.
Step 4: Distribute remaining electrons. Distribute the remaining valence electrons to satisfy the octet rule for each atom.
Detailed How-to: Drawing Each Bond
To draw the Lewis structure of H2SO4 comprehensively, follow these steps:
Step 1: Total valence electrons.
Here's the breakdown:
- Sulfur (S): 6 valence electrons
- Each Hydrogen (H): 1 valence electron × 2 = 2 valence electrons
- Each Oxygen (O): 6 valence electrons × 2 = 12 valence electrons
Total valence electrons: 6 (S) + 2 (2H) + 12 (2O) = 20 valence electrons
Step 2: Arrange the atoms.
Sulfur will be the central atom surrounded by two oxygen atoms and two hydrogen atoms:
Step 3: Form single bonds.
Draw single bonds between sulfur and each of the two oxygen atoms, and single bonds between sulfur and each of the two hydrogen atoms:
- S – O
- S – O
- S – H
- S – H
Step 4: Distribute remaining electrons.
Distribute the remaining 4 valence electrons as follows:
- Place two electrons (a pair) on each of the oxygen atoms to complete their octets.
- Place the remaining 2 electrons on the sulfur atom. This is acceptable since sulfur can have an expanded octet.
Step 5: Check octets and formal charges.
Ensure that each atom fulfills the octet rule where possible, and calculate any formal charges to ensure the structure is stable. Sulfur should have no formal charge, and each oxygen atom should have a formal charge of -1.
When you've done all this, your Lewis structure should look something like this:
O = S – O
H
H
Best Practices to Remember
Here are some best practices to keep in mind:
- Always double-check the central atom’s ability to expand its octet. Sulfur’s capacity to do so is vital for H2SO4.
- Focus on balancing formal charges to ensure the most stable structure.
- Use resonance structures if necessary to better describe the bonding.
Common Pitfalls and How to Avoid Them
Understanding and avoiding common mistakes can save you from confusion:
- Mistake: Using oxygen as the central atom due to its higher electronegativity.
- Solution: Remember that while oxygen is highly electronegative, it does not possess the ability to expand its octet like sulfur.
Another frequent error:
- Mistake: Neglecting to properly distribute the remaining valence electrons.
- Solution: Carefully distribute remaining electrons ensuring that each atom achieves the octet rule or duet rule where applicable.
Practical FAQ
Can I use resonance structures for H2SO4?
While the Lewis structure of H2SO4 does not typically require resonance structures, it’s important to understand that in some polyatomic molecules, resonance can help describe the bonding more accurately. For H2SO4, each oxygen can bond with sulfur through multiple resonance forms, delocalizing the charge to create a more stable structure. However, the primary Lewis structure you draw initially is accurate and sufficient for most purposes.
What’s the significance of the Lewis structure of H2SO4?
The Lewis structure of H2SO4 provides a visual representation of how electrons are shared and distributed across the molecule. This understanding is crucial for predicting molecular shape, reactivity, and bonding behaviors. Moreover, it forms a basis for learning about the properties of other molecules that have similar structures.
This guide offers a comprehensive, step-by-step method to draw the H2SO4 Lewis structure, combined with practical tips and best practices to avoid common mistakes. Whether you’re a student or a professional, mastering the Lewis structure of sulfuric acid will significantly enhance your understanding of molecular chemistry.