Understanding the H2C2 Lewis structure can be an intricate task but is essential for grasping basic principles in molecular chemistry. A Lewis structure provides a visual representation of the arrangement of atoms and the distribution of electrons within a molecule. This understanding is crucial for predicting the chemical behavior and stability of a molecule. In this guide, we delve into the specifics of the H2C2 Lewis structure, offering expert insights, practical examples, and actionable recommendations.
Key Insights
- The H2C2 molecule features a unique and relatively simple structure for learning the basics of molecular bonding.
- Proper placement of valence electrons is critical to accurately represent the molecular geometry and stability.
- Focus on optimizing the structure to minimize electron repulsion, adhering to the octet rule for stability.
Basic Composition and Molecular Geometry
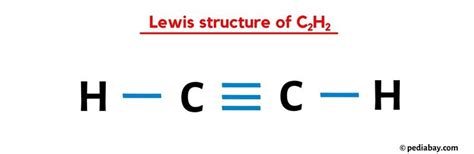
The H2C2 molecule comprises two hydrogen atoms (H) and one carbon atom ©. This molecular makeup allows us to explore fundamental concepts of chemical bonding. The carbon atom, with four valence electrons, forms covalent bonds with two hydrogen atoms. Each hydrogen atom shares its single valence electron with carbon, forming a single bond. The molecular geometry of H2C2 is linear, which means that the carbon atom is at the center, and the hydrogen atoms are positioned on either side. This linear structure is influenced by the sp hybridization of the carbon atom’s orbitals.Drawing the Lewis Structure
To construct the Lewis structure for H2C2, start by placing the carbon atom at the center, as it is the least electronegative element. Each hydrogen atom is then connected to the carbon by a single bond. The carbon atom shares its four valence electrons, one with each hydrogen. Following the octet rule, each hydrogen atom achieves a stable electron configuration with a single bond, as they do not require additional electrons. The structure can be represented as H-C=C-H, where the carbon-carbon bond is a double bond, which allows the carbon atoms to fulfill the octet rule more effectively.In practical applications, understanding the H2C2 Lewis structure is pivotal for studying larger hydrocarbons and learning about alkenes, where double bonds play a crucial role in reactivity and stability.
What is the significance of the double bond in H2C2?
The double bond in H2C2 significantly affects the molecule's reactivity and stability. It indicates a higher degree of electron density between the carbon atoms, making the molecule more reactive compared to single-bonded structures.
How does electron repulsion impact the H2C2 structure?
Electron repulsion in the H2C2 molecule is minimized by adopting a linear geometry. This arrangement ensures that the electrons around the central carbon atom are as far apart as possible, reducing repulsion and stabilizing the structure.
Mastering the Lewis structure of H2C2 provides foundational knowledge that can be applied to more complex molecules, emphasizing the importance of understanding bonding and molecular geometry.