Graphite is a fascinating form of carbon known for its exceptional physical and chemical properties. With applications ranging from pencils to advanced electronics, understanding its chemical formula and structure is pivotal for numerous scientific and industrial endeavors.
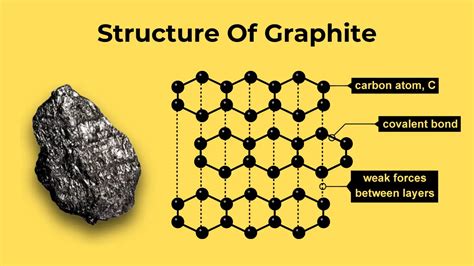
Graphite consists of layers of carbon atoms arranged in a hexagonal lattice. Each carbon atom in a graphite layer is bonded to three other carbon atoms by sp² hybridized orbitals, forming a two-dimensional planar structure. The layers are held together by weak van der Waals forces, enabling them to slide over each other, which imparts graphite its characteristic properties like its lubricating ability and ability to conduct electricity.
Key Insights
- Graphite’s chemical formula is C, signifying its elemental nature.
- Graphite’s structure comprises hexagonal layers of carbon atoms bonded by sp² hybridized orbitals.
- Interlayer van der Waals forces are crucial for graphite’s unique mechanical and electrical properties.
Chemical Formula and Elemental Composition
The chemical formula for graphite is simply C, reflecting its elemental carbon composition. Unlike diamond, another form of carbon with a tetrahedral crystal structure, graphite’s structure enables its unique properties. The carbon atoms in graphite are bound in planar layers, allowing each layer to conduct electricity and providing the material with its high thermal conductivity and its characteristic slippery feel when used as a lubricant.Structural Arrangement and Bonding
Graphite’s atomic structure consists of layers of carbon atoms arranged in a hexagonal close-packed lattice. Each carbon atom forms three sigma bonds with neighboring carbon atoms, resulting in each carbon having a planar structure. The fourth electron of each carbon atom forms a delocalized pi bond above and below the plane of the layer, contributing to graphite’s conductive properties. The layers stack in an ABAB sequence, with each layer offset from the adjacent one. These layers are bonded together by weak van der Waals forces, which enable them to slide past each other. This unique arrangement is responsible for graphite’s lubricating properties, making it an essential component in various mechanical applications.Real-World Applications
The properties of graphite make it indispensable in numerous applications. Its ability to conduct electricity, coupled with its chemical stability, make it a vital component in batteries, electrodes, and as a filler in various composite materials. Graphite’s lubricating properties are exploited in high-temperature settings, reducing friction and wear in mechanical parts. Its use in pencils, known as “lead,” is perhaps the most common application. Despite the name, modern “pencil lead” is actually made of graphite mixed with clay to achieve the desired hardness and writing properties.What distinguishes graphite from diamond?
Graphite and diamond both consist of carbon but differ in their atomic arrangements. In graphite, carbon atoms form layers in a hexagonal lattice, while in diamond, they form a three-dimensional network in a tetrahedral arrangement.
How is graphite used in industrial settings?
Graphite's unique properties make it an invaluable material in various industrial applications. It serves as an electrode in electrometallurgical processes, a lubricant in high-temperature mechanical components, and a conductive filler in composite materials.
Understanding the chemical formula and structure of graphite not only highlights its inherent properties but also underscores its versatility and importance across different industries. From everyday products like pencils to sophisticated technological applications, graphite’s unique characteristics continue to play a vital role in both current and future innovations.