The gold foil experiment, conducted by Ernest Rutherford in 1911, was a pivotal moment in the history of atomic science. This experiment led to the discovery of the atomic nucleus, revolutionizing our understanding of atomic structure. This guide is designed to not only explain the experiment in detail but also provide practical tips and solutions for anyone interested in exploring this fascinating subject.
Understanding the Gold Foil Experiment: A Solution to Fundamental Questions
The gold foil experiment stands as a cornerstone in the field of physics, addressing fundamental questions about the nature of matter. This experiment showed that atoms are mostly empty space, with a dense central core known as the nucleus. This revelation came as a surprise, as earlier models of the atom depicted a solar system-like structure where electrons orbited a central mass uniformly.
For those puzzled by these concepts, this guide offers a step-by-step journey through the experiment. It aims to demystify the intricate details and offer practical applications of the findings. By the end, you’ll not only understand the theory but also appreciate the implications for modern physics and beyond.
Quick Reference
Quick Reference
- Immediate action item: Familiarize yourself with basic atomic theory.
- Essential tip: Understand the role of alpha particles in the experiment.
- Common mistake to avoid: Misinterpreting the experiment’s results as a direct model of the atom.
The Setup: Detailed Step-by-Step Explanation
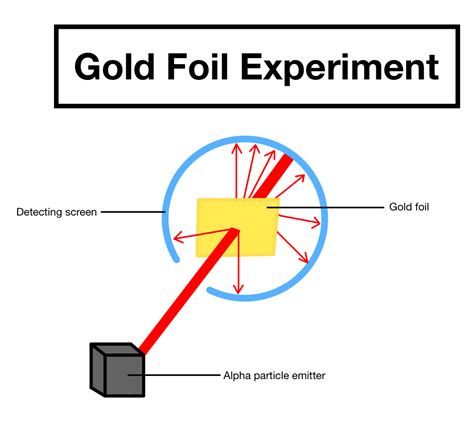
Let’s dive deeper into the setup of the gold foil experiment. To begin, Rutherford and his team took a thin sheet of gold foil and bombarded it with alpha particles. Alpha particles, which are essentially helium nuclei, were chosen for their dual benefits of high mass and positive charge, allowing them to interact strongly with atomic nuclei.
The experiment’s apparatus involved a radioactive source emitting alpha particles, a metal cylinder to focus the particles, and the gold foil. The setup was designed to allow a clear view of the scattering patterns produced when alpha particles interacted with the atoms in the foil.
Practical Tip: When setting up a similar experiment in a school lab, ensure you use a high-quality, ultra-thin gold foil for accurate results. Safety should also be a top priority, so handle radioactive sources with care, and always perform the experiment under supervision.
Observations and Analysis: What Did They Discover?
Rutherford and his team observed that most alpha particles passed through the foil with little to no deflection, consistent with the idea that atoms are mostly empty space. However, a small fraction of alpha particles were deflected at large angles, and a tiny fraction were reflected back. These observations led to a groundbreaking conclusion: atoms contain a dense central core.
Detailed Insight: The scattering pattern suggested that the positive charge and most of the mass of an atom were concentrated in a tiny nucleus, surrounded by a cloud of electrons. This model, later termed the Rutherford model or the “planetary model” of the atom, was a major step forward in atomic theory.
Best Practice: When discussing these findings, emphasize the unexpected nature of the observations and how they challenged existing atomic models. This helps students grasp the experimental nature of scientific discovery.
Implications of the Gold Foil Experiment: Advanced Understanding
The implications of Rutherford’s gold foil experiment are vast and profound. It not only overturned existing notions of atomic structure but also laid the groundwork for future discoveries in nuclear physics and quantum mechanics.
In modern physics, the experiment’s findings have led to a deeper understanding of nuclear forces and the behavior of subatomic particles. It paved the way for the development of atomic models that describe electrons in orbitals around the nucleus, leading to our current understanding of quantum mechanics.
Practical Application: For students and researchers, understanding the experiment’s implications can lead to a deeper appreciation of modern technologies, from medical imaging to nuclear power. It’s also a testament to how scientific experiments can lead to revolutionary changes in our understanding of the universe.
Practical FAQ: Addressing Common Questions
What is the significance of the gold foil experiment?
The gold foil experiment was significant because it provided concrete evidence that an atom has a small, dense nucleus surrounded by electrons. This finding was pivotal in overthrowing the plum pudding model of the atom and establishing the nuclear model of the atom, which is foundational to modern physics.
How can the results of the gold foil experiment be explained in simple terms?
Imagine shooting tiny, positively charged alpha particles at a very thin sheet of gold. Most of the particles go straight through, showing that atoms are mostly empty space. However, some particles bounce back or get deflected at large angles. This indicates that atoms have a tiny, dense center (the nucleus) where most of the atom’s mass is concentrated, surrounded by the electrons.
What were the main challenges faced during the gold foil experiment?
The main challenges included ensuring the foil was thin enough for the alpha particles to pass through, yet strong enough to avoid breaking apart during bombardment. There was also the challenge of precisely measuring the angles of deflection and accurately interpreting the scattering patterns. Safety in handling radioactive materials and ensuring minimal interference from other particles were also critical.
This guide has aimed to provide a thorough understanding of the gold foil experiment, its implications, and practical insights for both students and enthusiasts. By following this guide, you’ll gain a clear and comprehensive grasp of one of the most transformative experiments in the history of science.