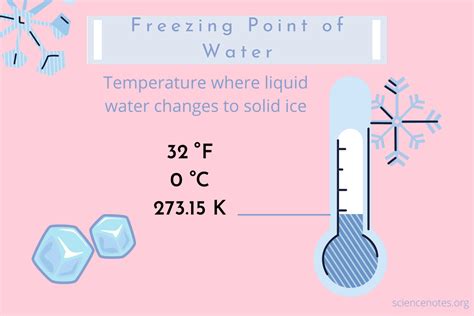
The exact freezing point of water in degree Celsius is a fundamental concept in various scientific disciplines, including chemistry, physics, and environmental science. The precise freezing point of water is 0°C, at which point it transitions from liquid to solid under standard atmospheric pressure. This knowledge is vital not only in scientific laboratories but also in practical applications such as meteorology, food preservation, and various industrial processes. Understanding this key fact allows professionals to make informed decisions that are grounded in scientific accuracy.
Key Insights
- The freezing point of water is a cornerstone in scientific measurement and practical applications.
- At 0°C, water transitions from liquid to ice under standard pressure, a phenomenon that must be understood for thermal management in various industries.
- Knowledge of the freezing point aids in ensuring accuracy in scientific experiments and industrial processes.
The Science Behind Water’s Freezing Point
The freezing point of water is defined as the temperature at which the equilibrium between the liquid and solid states of water occurs. This phenomenon is governed by thermodynamic principles and is pivotal for a myriad of scientific processes. At 0°C, water molecules cease their free motion in the liquid phase and arrange themselves in a crystalline structure, forming ice. This transition is not just a theoretical concept but has real-world implications. For instance, understanding this point is crucial in designing cooling systems and in studying climate change, where the behavior of ice caps and glaciers is of significant concern.Practical Applications in Industry and Daily Life
Knowledge of the exact freezing point of water plays a vital role in various sectors. In the food industry, for example, ensuring that water freezes at 0°C is essential for proper refrigeration and food preservation techniques. In pharmaceuticals, accurate temperature control is necessary for maintaining the efficacy of medications stored at specific temperatures. Additionally, meteorologists rely on this fact to predict weather patterns accurately, particularly those involving precipitation. The freezing point of water is a universal constant that serves as a reference point for many scientific and industrial applications.How does altitude affect the freezing point of water?
The freezing point of water can be affected by altitude due to changes in atmospheric pressure. At higher altitudes, where the pressure is lower, water can freeze at temperatures slightly below 0°C. This phenomenon can impact various natural processes and human activities, including the formation of ice at higher elevations.
Why is the exact freezing point of water important for scientists?
Scientists rely on the precise freezing point of water as a reference for calibration in thermometers and for creating controlled environments in experiments. This accurate measurement ensures the reliability of scientific data and the consistency of research outcomes.
Understanding the exact freezing point of water in degree Celsius is not just an academic exercise but a practical necessity. It underpins various scientific and industrial methodologies, helping professionals to make informed decisions and conduct accurate research. Whether it’s in environmental science, industrial applications, or everyday tasks, knowing this fundamental fact is indispensable.