Understanding the enthalpy of fusion can be an eye-opener for anyone interested in thermodynamics and material science. This guide will take you step-by-step through the fundamental concepts, practical applications, and hidden secrets of the enthalpy of fusion. Our aim is to arm you with actionable knowledge that can be directly applied in both academic and real-world scenarios.
What Is Enthalpy of Fusion?
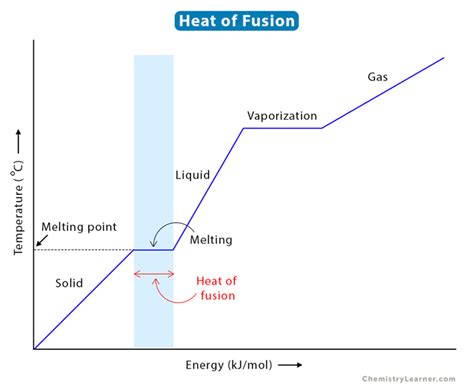
Enthalpy of fusion refers to the amount of energy required to change a substance from a solid to a liquid at its melting point without changing temperature. This energy is crucial for understanding phase changes in materials and plays a vital role in processes ranging from cryogenics to metallurgy.Problem-Solution Opening: Addressing User Needs
If you’ve ever wondered how much energy is involved in melting a block of ice or why certain materials require specific amounts of heat to melt, this guide is for you. Often, understanding these details can be perplexing, but with the right approach, you’ll be able to grasp the essential concepts and apply them to solve real-world problems. Whether you’re working on a physics project, a chemical engineering task, or simply trying to better understand a science concept, this guide offers a detailed exploration of enthalpy of fusion, from basic principles to advanced applications.Quick Reference
Quick Reference
- Immediate action item: Calculate the enthalpy of fusion for a substance by multiplying its molar mass by its specific fusion enthalpy.
- Essential tip: Use this formula: ( Q = n \times \Delta H_f ), where ( Q ) is the heat required, ( n ) is the number of moles, and ( \Delta H_f ) is the enthalpy of fusion.
- Common mistake to avoid: Confusing enthalpy of fusion with latent heat of vaporization, which applies to transitions from liquid to gas.
Deep Dive into Enthalpy of Fusion
To fully understand enthalpy of fusion, let’s break down the science behind it and how it can be practically applied.The Science Behind Enthalpy of Fusion
When a substance melts, it absorbs heat energy. This energy breaks the bonds that hold the solid’s particles in a fixed structure, allowing them to move more freely as a liquid. The amount of energy needed for this transition is its enthalpy of fusion. The process can be understood through these steps:- Bond Breaking: Energy is used to break intermolecular bonds, transforming solid molecules into liquid molecules.
- Temperature Equilibrium: During the phase change, temperature remains constant at the melting point until all the solid has melted.
- Energy Absorption: The absorbed energy is stored in the substance until the phase change is complete.
Practical Calculations: How to Determine Enthalpy of Fusion
Determining the enthalpy of fusion involves a straightforward yet precise calculation. Follow this step-by-step guide to understand the process.Step 1: Identify the substance and its molar mass.
Step 2: Find the specific enthalpy of fusion ( \Delta H_f ) for that substance from reliable sources such as scientific literature or databases.
Step 3: Calculate the number of moles ( n ) of the substance if you're dealing with a specific mass.
Step 4: Use the formula Q = n \times \Delta H_f to calculate the required heat (enthalpy of fusion).
- Example: Suppose you have 10 grams of water (molar mass 18 g/mol) and the specific enthalpy of fusion for water is 334 J/g. Calculate the number of moles: n = \frac{10 \text{ g}}{18 \text{ g/mol}} = 0.56 \text{ mol} .
- Use the formula: Q = 0.56 \text{ mol} \times 334 \text{ J/g} \times 18 \text{ g/mol} = 3340.8 \text{ J} .
Advanced Applications: Using Enthalpy of Fusion in Real-World Scenarios
In practical applications, enthalpy of fusion is used in various fields like material science, environmental science, and even in culinary arts. Let’s look at some examples:- Industrial Applications: In metallurgy, understanding the enthalpy of fusion of metals is crucial for optimizing the melting process, ensuring energy efficiency, and preventing material wastage.
- Environmental Science: When studying climate change, scientists use enthalpy of fusion to understand phase changes in ice caps and glaciers, contributing to models predicting global warming impacts.
- Culinary Science: For instance, the enthalpy of fusion for fats is used in understanding cooking temperatures for different meats, ensuring precise cooking methods.
Practical FAQ Section
What is the difference between enthalpy of fusion and latent heat of vaporization?
The enthalpy of fusion is the energy required to convert a solid into a liquid at its melting point, keeping the temperature constant. In contrast, the latent heat of vaporization is the energy needed to convert a liquid into a gas at its boiling point, again without changing temperature. While both are related to phase changes, they apply to different transitions.
Common Mistakes to Avoid
Understanding enthalpy of fusion is made simpler if you avoid some common pitfalls:- Don't confuse it with latent heat of vaporization.
- Ensure accurate measurements for the substance's molar mass and specific enthalpy values.
- Keep the calculations straightforward, using reliable data sources for enthalpy values.
With this guide, you now have the foundation and practical tools to delve into the enthalpy of fusion and apply it in a variety of contexts. Whether it's for academic purposes or professional application, this knowledge will prove invaluable in your understanding of thermodynamics and phase changes.
Keep experimenting, stay curious, and always strive to apply these concepts to real-world problems. Understanding the enthalpy of fusion not only demystifies the melting process but also equips you with critical insights into the broader realm of physical sciences.