Imagine you’re trying to decide between two types of activities: one that needs a lot of energy to get going but continues on its own, and another that runs smoothly and requires only a small push to keep it going. This is akin to understanding the distinction between endergonic and exergonic processes. If you’ve ever felt confused about these terms in the context of chemistry, biology, or even simple daily activities, this guide will break it down in a way that’s straightforward and easy to grasp. We’ll address your questions, provide practical examples, and offer actionable advice to make this clearer for you.
Understanding Endergonic and Exergonic Processes
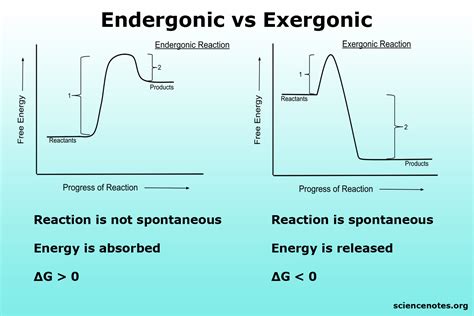
Endergonic and exergonic processes are fundamental concepts in thermodynamics, essential for understanding how energy flows in biological and chemical systems. Let's break down what these terms mean in simple, practical terms. An endergonic process requires an input of energy to proceed. This means that the reaction needs an initial push to get started, and it usually ends up in a higher energy state than it started. Think about it like climbing a hill – you expend energy to get to the top, and once you start going down, you maintain that momentum on your own. On the other hand, an exergonic process releases energy as it proceeds. This means that once initiated, the reaction can often continue on its own without needing additional input, and it usually results in a lower energy state. This is similar to descending a hill, where the energy you used to climb up can propel you down without additional effort.
These processes are not just abstract concepts; they play a critical role in everyday life, from the way our bodies function to the chemical reactions that occur in everyday cooking or cleaning.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: To quickly understand the difference, think of endergonic processes like climbing a hill (requires energy input) and exergonic processes like descending a hill (releases energy).
- Essential tip with step-by-step guidance: To identify if a process is endergonic or exergonic, observe whether energy is required to start the process or if it naturally releases energy.
- Common mistake to avoid with solution: Confusing endergonic with exergonic processes. To avoid this, remember that energy input signifies endergonic, while energy release signifies exergonic.
Detailed Explanation: Endergonic Processes
Endergonic processes are essential for building complex structures and storing energy. They require an input of energy to proceed. Here's a step-by-step breakdown to understand these processes better:
- Identify the process: Determine if the process is synthesizing something or building a structure. For instance, the synthesis of proteins from amino acids is an endergonic process because it requires energy input.
- Understand the energy requirement: Note that these processes often involve chemical reactions where the formation of bonds requires an energy input. For example, photosynthesis in plants is an endergonic reaction because it requires sunlight energy to convert carbon dioxide and water into glucose and oxygen.
- Recognize the outcome: Endergonic processes usually result in a higher energy state than the starting materials. This means that the products of the reaction have more energy than the reactants. For instance, the formation of glucose from carbon dioxide and water has a higher energy content than the original reactants.
Here's a practical example to make this clearer:
- Cooking an egg: Boiling an egg is an endergonic process because the heat energy required to cook it has to be supplied from an external source. The proteins in the egg denature and form new bonds, resulting in a higher energy state compared to raw egg proteins.
Detailed Explanation: Exergonic Processes
Exergonic processes release energy, which makes them more spontaneous. These processes typically break down complex structures or substances into simpler forms. Here's a step-by-step guide to understanding exergonic processes:
- Identify the process: Determine if the process involves breaking down something. For example, the breakdown of glucose into carbon dioxide and water is an exergonic process because it naturally releases energy.
- Understand the energy release: Note that these processes often involve chemical reactions where the breaking of bonds releases energy. For instance, cellular respiration in organisms is an exergonic reaction because it releases energy stored in glucose to produce ATP, carbon dioxide, and water.
- Recognize the outcome: Exergonic processes usually result in a lower energy state than the starting materials. This means that the products of the reaction have less energy than the original reactants. For example, the breakdown of glucose has a lower energy content than the original glucose molecule.
Here's a practical example to make this clearer:
- Metabolism: When your body breaks down a piece of toast, the exergonic process releases energy that your body can use for various functions, including movement and maintaining body temperature. This is a perfect example of energy release from a higher energy state (the toast) to a lower energy state (CO₂ and water).
Practical FAQ
How can I apply this knowledge to understand biological processes?
Applying your understanding of endergonic and exergonic processes can greatly enhance your grasp of biological functions. For instance, consider the process of muscle contraction. When you exercise, your muscles undergo exergonic reactions to break down ATP (adenosine triphosphate), releasing energy needed for muscle contraction. Conversely, the regeneration of ATP from ADP (adenosine diphosphate) and inorganic phosphate is an endergonic process that requires energy input, often supplied by food.
Similarly, you can understand cellular processes like photosynthesis and respiration. Photosynthesis is an endergonic process where plants convert sunlight into chemical energy (glucose). In contrast, cellular respiration is an exergonic process where cells break down glucose to release energy for cellular activities. Recognizing these processes helps you understand how energy flows within biological systems.
Practical Application and Examples
To make these concepts even more practical, let's look at real-world applications:
- Building and breaking down structures: Imagine you are building a house. The construction process requires energy to put materials together (endergonic). Conversely, if you tear down the house, the demolition process releases energy as materials are broken apart (exergonic).
- Everyday cooking: Cooking involves many endergonic processes, like boiling an egg or baking a cake, which require energy input. Conversely, processes like the melting of ice cream (as it melts in your hand) are exergonic because they naturally release energy as ice cream transitions to a liquid state.
Incorporating these concepts into your daily life can help you recognize and appreciate the energy dynamics at play in both natural and human-made processes.
Best Practices for Understanding Energy Processes
Here are some best practices to deepen your understanding of endergonic and exergonic processes:
- Observe everyday activities: Pay attention to the energy requirements and outcomes of daily activities. Notice which processes require energy input and which release energy.
- Use analogies: Employ relatable analogies to simplify complex concepts. For instance, compare endergonic processes to building something from scratch and exergonic processes to dismantling a structure.
- Practice with examples: Regularly practice identifying and categorizing processes as endergonic or exergonic. Use examples from both biology and everyday life to strengthen your understanding.
By following these practices, you can develop a more intuitive grasp of how energy flows in various processes.
Conclusion
Understanding the differences between endergonic and exergonic processes is fundamental for grasping how energy operates in both biological and chemical systems. By breaking down these concepts into practical examples and daily activities, you can see how they relate to everyday life. Whether you’re cooking, exercising, or studying cellular processes, this knowledge equips you with the insights to appreciate the energy dynamics at play. With practice and real-world application, these concepts will become second nature,