Understanding the difference between electrolytic and voltaic cells is crucial for anyone delving into the field of electrochemistry. These two types of cells serve different purposes and work on entirely different principles. This guide will walk you through the distinctions, practical applications, and essential tips to make your study of electrochemistry more fruitful.
Introduction to Electrolytic and Voltaic Cells
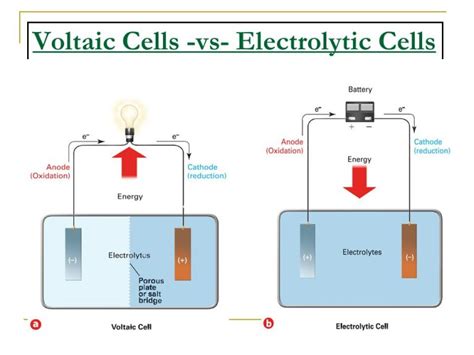
When it comes to the study of electrochemistry, cells are a fundamental concept. Whether you’re a student, an engineer, or a hobbyist, grasping the functionality and application of electrolytic and voltaic cells can significantly enhance your understanding of chemical reactions and energy storage. The key difference lies in their applications: electrolytic cells drive non-spontaneous reactions through electrical energy, whereas voltaic cells generate electrical energy from spontaneous chemical reactions.
Electrolytic cells are used to drive chemical changes that wouldn’t occur naturally, while voltaic cells (often referred to as galvanic cells) generate an electric current from a spontaneous redox reaction. Misunderstanding these differences can lead to incorrect applications and ineffective outcomes, which is why a thorough guide is necessary to clarify these concepts.
Quick Reference Guide
Quick Reference
- Immediate action item with clear benefit: Begin by identifying whether your project requires energy generation or consumption. This decision will guide you to use the appropriate cell type.
- Essential tip with step-by-step guidance: To build a voltaic cell, gather zinc and copper electrodes, dilute sulfuric acid, and connect these components to form a circuit. For an electrolytic cell, obtain a direct current power source and appropriate electrolytes.
- Common mistake to avoid with solution: Avoid confusing the roles of anode and cathode in both types of cells. In a voltaic cell, the zinc electrode (anode) loses electrons, whereas in an electrolytic cell, the cathode gains electrons. Misunderstanding these roles can lead to incorrect cell configuration.
Detailed How-To Sections
Electrolytic Cells: Principles and Applications
Electrolytic cells are used to perform non-spontaneous chemical reactions that require an input of electrical energy. These cells are particularly useful in applications like electrolysis, which is used in refining metals, plating metals, and synthesizing compounds. Here’s how you can understand and work with electrolytic cells:
Building an Electrolytic Cell
To construct an electrolytic cell, you’ll need a few key components:
- Electrolyte: A conductive solution or molten compound that facilitates the movement of ions.
- Anode and Cathode: Typically made from inert materials like platinum or graphite.
- Direct current (DC) power supply.
Here’s a step-by-step guide:
Step 1: Setup the Electrolyte
Choose your electrolyte carefully based on the reaction you want to facilitate. For example, for metal plating, an acidic solution like sulfuric acid is often used.
Step 2: Insert Electrodes
Place your inert anode and cathode into the electrolyte. Make sure there’s sufficient space between them for efficient ion movement.
Step 3: Connect to Power Supply
Connect the anode to the negative terminal and the cathode to the positive terminal of the DC power supply. The power supply should provide enough voltage to drive the desired reaction.
Step 4: Monitor the Process
Start the process and monitor the reaction. Electrolysis reactions can produce gas bubbles at the electrodes, so always ensure safe handling.
Voltaic Cells: Principles and Applications
In contrast to electrolytic cells, voltaic cells generate electrical energy from spontaneous redox reactions. They are the backbone of most modern batteries and power sources.
Building a Voltaic Cell
To create a voltaic cell, you’ll need:
- Two different metals with different electrode potentials.
- Electrolyte solution, often an acid or salt solution.
- Insulating material to connect the two metals.
Step 1: Gather Components
Common metals used in voltaic cells include zinc and copper. For the electrolyte, a dilute sulfuric acid solution is typically employed.
Step 2: Assemble the Cell
Place the zinc electrode in one beaker containing the electrolyte, and the copper electrode in another. Connect them with an insulating bridge filled with the electrolyte solution.
Step 3: Close the Circuit
Connect the two electrodes with a wire to form a closed circuit, allowing the electrons to flow from the zinc anode to the copper cathode through the external circuit.
Step 4: Observe the Reaction
As the reaction progresses, electrons flow from the zinc electrode into the copper electrode, generating a flow of electrical current.
Practical FAQ
What’s the difference between an electrolytic and voltaic cell?
An electrolytic cell uses electrical energy to drive non-spontaneous reactions, while a voltaic cell (galvanic cell) generates electrical energy from spontaneous redox reactions. Electrolytic cells are used in processes like electrolysis, whereas voltaic cells are used in batteries and other power sources.
Why is it important to understand the role of the anode and cathode?
Understanding the roles of the anode and cathode is crucial because it determines the direction of electron flow and ion movement in both electrolytic and voltaic cells. In electrolytic cells, the cathode is positively charged and attracts negatively charged ions, while the anode is negatively charged and releases electrons. In contrast, in voltaic cells, the zinc anode (negative) loses electrons, and the copper cathode (positive) gains electrons. Misunderstanding these roles can lead to incorrect cell setups and failure to achieve desired outcomes.
How do I choose the right electrolyte for my electrolytic cell?
The choice of electrolyte depends on the specific reaction you want to carry out. For metal plating, acidic solutions like sulfuric or hydrochloric acid are common. For other applications, the electrolyte should have suitable ions to facilitate the reaction. Always ensure the electrolyte is compatible with your electrodes and the power supply used.
Mastering the distinctions and applications of electrolytic and voltaic cells will not only deepen your understanding of electrochemistry but also equip you with practical skills for various scientific and engineering projects.