Democritus Atomic Theory: The Ancient Breakthrough
Democritus, a Greek philosopher born around 460 BCE, introduced one of the first philosophical theories of matter. His ideas have influenced the development of modern atomic theory, which is essential for our understanding of chemistry and physics today. This guide will walk you through the key concepts and advancements that Democritus proposed, using clear examples and practical insights to help you grasp this foundational ancient breakthrough.
Understanding Democritus’ Atomic Theory
Democritus proposed that all matter is composed of small, indivisible particles called atoms. His ideas were groundbreaking because they offered a systematic approach to understanding the composition of matter, long before modern science could validate or refine these concepts.
Problem-Solution Opening
For centuries, the nature of matter perplexed scholars and philosophers. Democritus addressed this ancient dilemma by proposing that everything in the universe is made up of tiny, indivisible units. His atomic theory offered a simple yet profound explanation for the complexity of physical substances. Today, as we study the building blocks of matter, the fundamental concept remains: everything is made of atoms. This guide will explore Democritus' theory, examining his contributions and how they laid the groundwork for modern atomic theory.
Quick Reference
Quick Reference
- Immediate action item: Research the historical context of Democritus’ theory to understand how it shaped modern science.
- Essential tip: Understand that Democritus proposed that all matter is made of indivisible atoms, which is a fundamental concept in modern chemistry.
- Common mistake to avoid: Confusing Democritus' philosophical ideas with modern scientific atomic theory. Note that while his ideas weren't scientifically accurate, they laid the conceptual groundwork.
The Concept of Atoms
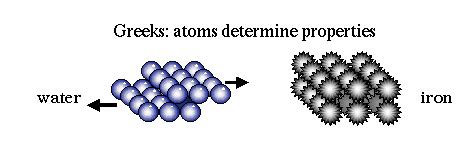
Democritus envisioned atoms as the basic units of matter that were indivisible and indestructible. He posited that different physical properties of substances were due to the variations in the size, shape, and arrangement of these atoms. Despite the philosophical nature of his theory, it was a radical departure from the prevailing idea that matter could be divided indefinitely.
The Four Fundamental Properties
Democritus described atoms as having four fundamental properties: shape, size, position, and motion. He believed that the differences in these properties determined the different types of matter. While modern science has expanded on this idea by identifying many more characteristics of atoms, his foundational concepts are still relevant today.
Atoms and Void
According to Democritus, atoms and the void (empty space) around them were the only true components of the universe. He theorized that when atoms combined in different ways, they formed the variety of objects we observe in the world. This idea of the void being an essential part of the universe was another significant aspect of his theory, which has echoes in modern scientific concepts like space in the quantum realm.
How Democritus’ Ideas Influenced Modern Science
Although Democritus’ ideas were not scientifically proven during his lifetime, they provided a framework for future scientific inquiry. The atomic theory proposed by Democritus influenced subsequent thinkers and laid the groundwork for the development of atomic theory in the modern era.
Rediscovery of Atomic Theory
It wasn’t until the 17th and 18th centuries that the concept of atoms was revived and refined. Scientists like John Dalton furthered the atomic theory, providing empirical evidence that supported Democritus' foundational ideas. Dalton’s atomic theory proposed that elements were composed of atoms that could combine to form compounds, a crucial step toward the modern understanding of chemistry.
Modern Atomic Theory
The principles introduced by Democritus led to the development of the modern atomic theory, which describes atoms as consisting of subatomic particles like protons, neutrons, and electrons. This theory provides a comprehensive understanding of matter at the atomic and molecular levels, which is essential for fields such as chemistry, physics, and materials science.
Practical Applications of Atomic Theory
Understanding atomic theory is not just a theoretical exercise; it has practical applications in various scientific fields. From developing new materials to understanding the fundamental processes of life, atomic theory has immense practical value.
Application in Chemistry
In chemistry, the understanding of atoms and their interactions is fundamental. Atomic theory explains how different elements combine to form compounds and how these compounds interact in chemical reactions. This knowledge is critical for fields like pharmaceuticals, materials science, and environmental science.
Application in Physics
In physics, atomic theory is the basis for understanding and describing the behavior of particles. Concepts like quantum mechanics, which describe the behavior of atoms and subatomic particles, have led to technological advancements like semiconductors, which are the building blocks of modern electronics.
Frequently Asked Questions (FAQ)
What are the main components of Democritus’ atomic theory?
Democritus’ atomic theory posits that all matter is composed of tiny, indivisible units called atoms. He suggested that atoms differ in shape, size, and arrangement, leading to the variety of matter observed. Although his ideas were philosophical rather than scientific, they provided the conceptual framework for future atomic theory.
How did Democritus’ theory influence modern science?
Although not scientifically validated in Democritus’ time, his theory laid the conceptual groundwork for atomic theory. It influenced later thinkers like John Dalton, who developed a more empirical atomic theory. This, in turn, contributed to the modern atomic theory that forms the foundation of chemistry and physics.
What are the practical applications of atomic theory today?
Atomic theory has numerous practical applications, especially in chemistry and physics. In chemistry, it helps in understanding chemical reactions and developing new materials. In physics, it is fundamental to quantum mechanics and the development of modern technology, such as semiconductors in electronics.
Democritus’ atomic theory, though simplistic by today’s standards, was a revolutionary concept that significantly influenced the development of modern atomic theory. By exploring the foundational ideas he proposed, we can appreciate the profound impact he had on science. This ancient breakthrough, rooted in philosophical thought, set the stage for centuries of scientific discovery and continues to underpin our modern understanding of the material world.