Democritus is frequently credited with proposing one of the earliest atomic theories in Western philosophy. While his model of the atom is vastly simplified compared to modern atomic theory, it laid a fundamental groundwork for future scientific discoveries. Democritus posited that everything in the universe is composed of atoms, indivisible and indestructible particles that float through the void, interacting to form all matter and phenomena. This concept was revolutionary for its time, challenging the prevalent theories of his era.
Key Insights
- Democritus' atom theory suggested that all matter is composed of indivisible particles.
- He believed that differences in physical properties are due to variations in the shape, size, and arrangement of these atoms.
- While simplistic, this idea was revolutionary for its time and influenced later atomic theories.
This section explores the historical context and evolution of Democritus’ atomic model, underscoring its importance in the history of science.
Understanding Democritus’ atomic theory begins with recognizing his departure from the conventional wisdom of his time. The prevalent thought held that matter was composed of classical elements—earth, water, air, and fire—which were perceived as fundamental and unchangeable. Democritus challenged this notion by suggesting that these elements could be broken down into smaller, indivisible units. This notion of indivisibility laid the foundation for modern atomic theory, though Democritus did not have empirical evidence to support his claims. Instead, he based his theory on logical reasoning and philosophical arguments, asserting that atoms must exist because matter could not be infinitely divisible.
Democritus’ Model and Its Impact
Democritus’ model proposed that atoms were eternal, indestructible, and varied in shape, size, and arrangement. According to him, the diversity in the physical world stemmed from these variations among atoms. This idea was groundbreaking because it proposed a mechanistic explanation for natural phenomena, a concept far removed from mythological or supernatural interpretations. Though his atomic theory lacked experimental validation, it planted the seeds for later scientific exploration.
In his theory, Democritus illustrated that different physical properties like color, texture, and density could be attributed to the unique configurations of these atoms. This insight was a significant shift towards the understanding of matter as having a structured and definable composition. Although Democritus did not have the tools or knowledge to test his ideas, his conceptual framework inspired future thinkers like Lucretius, who expanded on Democritus’ atomic model with a more detailed mechanistic explanation in his epic poem “De Rerum Natura.”
Legacy of Democritus' Atom Theory
The legacy of Democritus’ atom model is profound. While his ideas were rudimentary, they provided a philosophical basis for the atomic hypothesis that would later be rigorously tested and confirmed by scientists such as John Dalton in the 19th century. Dalton’s atomic theory, which introduced quantitative measures to atomic theory, marked a significant departure from Democritus’ original concepts but acknowledged the ancient philosopher’s foundational contribution.
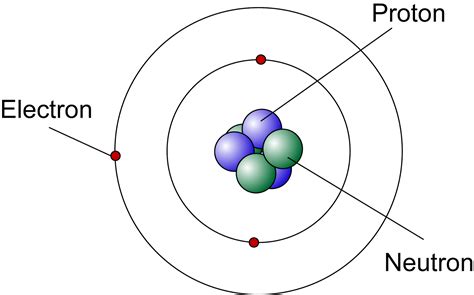
Moreover, Democritus’ emphasis on the indivisibility of atoms paralleled later discoveries in physics and chemistry. The concept of atoms as the fundamental units of matter would lead to the eventual discovery of subatomic particles like protons, neutrons, and electrons, culminating in our modern understanding of atomic structure.
Was Democritus' atomic theory validated during his lifetime?
No, Democritus' atomic theory was not empirically validated during his time. His ideas were primarily philosophical and based on logical reasoning rather than experimental evidence.
How did Democritus' atomic model influence modern science?
Democritus' atomic model laid the philosophical groundwork for the atomic theory, influencing later scientists. His ideas about indivisibility and the existence of a void provided the foundational concepts that were later refined and validated through rigorous experimentation.
This thorough analysis of Democritus’ atomic model highlights its revolutionary impact and enduring legacy in the realm of scientific thought. By proposing the idea of atoms as the building blocks of matter, Democritus significantly advanced the intellectual landscape, setting the stage for the empirical atomic theories that followed.