Understanding the Polarity of CS2: A Comprehensive Guide
Have you ever wondered whether carbon disulfide (CS2) is polar or nonpolar? Understanding the polarity of molecules like CS2 is critical in chemistry because it influences many properties of compounds, including solubility, melting and boiling points, and reactivity. This guide is designed to break down the concepts in a step-by-step manner with practical examples, real-world applications, and expert tips to ensure you grasp these fundamental chemistry concepts effectively.
Why Polarity Matters
Polarity refers to the distribution of electrical charge over the atoms joined by the bond. In simple terms, it describes whether the electrons are shared equally or not between the bonded atoms. Polar molecules have an uneven distribution of electron density, resulting in a net dipole moment. Nonpolar molecules, on the other hand, have an even distribution of electron density, resulting in no net dipole moment. This distinction is crucial because it determines how a molecule will interact with others and, therefore, its physical properties.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Sketch the Lewis structure of CS2 to understand its molecular geometry.
- Essential tip with step-by-step guidance: Use VSEPR (Valence Shell Electron Pair Repulsion) theory to predict the molecular shape and then determine the polarity.
- Common mistake to avoid with solution: Assuming linear geometry leads to polarity; ensure you use the correct molecular geometry to assess polarity correctly.
Now that we’ve established the importance of understanding molecular polarity, let’s dive into the specifics of CS2. This molecule will be our focal point to explore the practical steps and theoretical understanding required to unravel its chemical mystery.
Determining the Polarity of CS2
To determine whether CS2 is polar or nonpolar, we need to follow several steps methodically. Here’s a thorough guide to help you understand the process.
Step 1: Draw the Lewis Structure
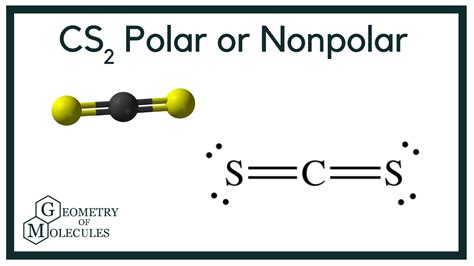
The first step in determining polarity is to draw the Lewis structure of the molecule. CS2 has one carbon atom (C) bonded to two sulfur atoms (S). Here’s how you do it:
- Carbon has 4 valence electrons.
- Each sulfur atom has 6 valence electrons.
- Carbon will form double bonds with each sulfur atom.
- Distribute the remaining electrons to satisfy the octet rule.
The Lewis structure for CS2 will show one carbon atom at the center with double bonds extending out to each sulfur atom. The structure will appear as:
S = C = S
Step 2: Predict the Molecular Geometry
Using the VSEPR theory, we can predict the molecular geometry. CS2 has two regions of electron density around the central carbon atom—each sulfur atom contributes one region. Since there are no lone pairs on the carbon atom, the geometry is linear.
Step 3: Determine Polarity Based on Geometry
For molecules with a linear geometry, polarity depends on the electronegativity of the atoms involved. Carbon is less electronegative than sulfur. However, due to the symmetrical arrangement of the two sulfur atoms around the central carbon, any dipole moment generated by one sulfur atom is canceled by the equal and opposite dipole moment of the other sulfur atom.
Therefore, despite the electronegativity difference, CS2 is nonpolar. The symmetrical linear shape means that the dipole moments cancel each other out completely.
Tips, Best Practices, and Practical Examples
Understanding the steps to determine molecular polarity is not just an academic exercise—it has real-world applications. For example, knowing that CS2 is nonpolar helps in understanding its behavior in various solvents. Let’s delve into some best practices and practical examples to cement these concepts.
Practical Tip 1: Always Sketch the Lewis Structure
Before jumping to conclusions about polarity, sketch the Lewis structure. This fundamental step helps ensure you understand the electron distribution and bond formation, which is critical in determining polarity.
Practical Tip 2: Use VSEPR Theory to Predict Geometry
The VSEPR theory is a powerful tool for predicting the geometry of molecules. Make it a habit to apply this theory to visualize the molecular shape before assessing polarity.
Practical Tip 3: Consider Symmetry
Symmetry plays a vital role in determining molecular polarity. If a molecule has a symmetrical arrangement of atoms, even if the individual bonds are polar, the molecule may be nonpolar. Always check for symmetry.
Let’s apply these tips with a practical example:
Example: Compare Polarity of CS2 and CO2
Both CS2 and CO2 are linear molecules. CS2 is nonpolar due to its symmetrical structure. CO2, however, has a linear geometry as well but is composed of two polar C=O bonds. Even though each bond is polar, the molecule remains nonpolar due to its linear and symmetrical shape. Understanding these nuances helps in grasping the concept of polarity and its implications in chemical interactions.
Frequently Asked Questions
Why is CS2 nonpolar despite the electronegativity difference?
The electronegativity difference between carbon and sulfur indicates that the C=S bonds are polar. However, the linear and symmetrical structure of CS2 causes the dipole moments to cancel each other out. This symmetry overrides the effect of the individual bond polarities, resulting in a nonpolar molecule.
How does the polarity of a molecule affect its physical properties?
Polarity significantly affects physical properties such as boiling and melting points, solubility, and reactivity. Polar molecules generally have higher boiling and melting points compared to nonpolar molecules of similar size due to stronger intermolecular forces. They also tend to dissolve better in polar solvents like water.
Understanding the polarity of molecules like CS2 is not only essential for grasping basic chemistry concepts but also for predicting and explaining their behavior in various chemical environments. By following the steps outlined in this guide and applying practical examples and tips, you can develop a robust understanding of molecular polarity.
Conclusion
The journey to uncovering the polarity of CS2 demonstrates the interplay between molecular structure and chemical properties. By systematically analyzing the Lewis structure, molecular geometry, and symmetry, you can confidently determine whether a molecule is polar or nonpolar. This knowledge is not just theoretical but has practical implications in various scientific fields, from chemistry to materials science. Continue to practice these concepts with different molecules to deepen your understanding and proficiency.
Remember, the key to mastering chemistry lies in a solid foundation of fundamental concepts coupled with practical application and critical thinking. Happy studying!