In the realm of chemistry, concentration is a fundamental concept that underpins many processes and reactions. Understanding concentration isn’t just a theoretical exercise; it has practical implications for everything from pharmaceuticals to environmental science. The idea revolves around the amount of a substance present in a given volume or area, which is pivotal for both laboratory work and real-world applications.
This article aims to clarify the concept of concentration in chemistry, ensuring beginners grasp the essentials without the complexity. We will delve into practical insights and real-world examples to illustrate this vital topic.
Key Insights
- Primary insight with practical relevance: Concentration is a measure of how much of a substance is present in a given volume or area, which is critical for determining the effectiveness of solutions in both laboratory experiments and real-world applications.
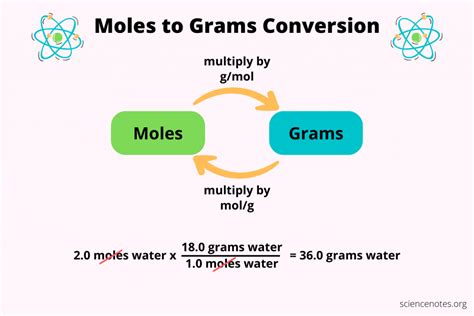
- Technical consideration with clear application: The molarity (M) of a solution is a common concentration measurement in chemistry, expressed as the number of moles of solute per liter of solution. This measurement is vital for balancing chemical equations and understanding reaction rates.
- Actionable recommendation: When preparing solutions, always ensure precise measurements of solutes and solvents to maintain the desired concentration for your experiment.
Understanding the Basics of Concentration
Concentration in chemistry refers to the amount of a substance (solute) dissolved in a solvent to form a solution. There are various ways to express concentration, such as molarity, molality, and percentage concentration, each suitable for different scenarios.
Molarity (M) is perhaps the most common way to express concentration. It defines the number of moles of solute per liter of solution (moles/L). This is essential in reactions where the volume of the solution directly influences the reaction rate. For example, in a titration, the precise molarity of the titrant is crucial for accurate determination of the analyte’s concentration.
Molality (m) is another concentration measurement, defined as the number of moles of solute per kilogram of solvent. It’s particularly useful in colligative property calculations where the solvent’s mass is more relevant than its volume, such as in boiling point elevation or freezing point depression.
Percentage concentration, typically expressed as percentage by mass or percentage by volume, is also common, especially in industries like pharmaceuticals. It indicates the amount of solute per 100 grams or 100 milliliters of solution.
Concentration in Real-World Applications
Understanding concentration extends beyond the lab into practical applications. In medicine, for instance, the concentration of active ingredients in drugs is vital for efficacy and safety. Too low a concentration might render the drug ineffective, while too high a concentration could be toxic.
In environmental science, concentration measurements are crucial for monitoring pollutants in water and air. Determining the concentration of harmful substances like heavy metals or chemicals helps assess environmental safety and compliance with regulatory standards.
A practical example is the preparation of buffer solutions in laboratories. Buffers maintain a constant pH, crucial for biological systems. By carefully adjusting the concentration of acids and bases, chemists can create solutions that resist changes in pH, essential for numerous biochemical processes.
Why is the concept of concentration important in laboratory experiments?
The concentration of solutions is crucial in determining the outcomes of chemical reactions. Accurate concentration measurements ensure that reactions proceed as expected, helping to predict reaction rates, product yields, and even the stability of solutions.
How does concentration relate to the effectiveness of cleaning agents?
In cleaning solutions, concentration directly affects the cleaning power. A higher concentration of a cleaning agent, such as bleach, often results in a more effective cleaning process, capable of breaking down and removing more dirt and stains.
In conclusion, concentration is a critical concept in chemistry with extensive applications in various fields. From ensuring the efficacy of medical treatments to monitoring environmental pollutants, understanding and accurately measuring concentration is essential for both theoretical and practical pursuits. By mastering these principles, beginners can lay a solid foundation for more advanced chemical studies.